Improving Australian National Bowel Cancer Screening Program outcomes through increased participation and cost-effective investment
- PMID: 32012174
- PMCID: PMC6996821
- DOI: 10.1371/journal.pone.0227899
Improving Australian National Bowel Cancer Screening Program outcomes through increased participation and cost-effective investment
Abstract
Background: The Australian National Bowel Cancer Screening Program (NBCSP) provides biennial immunochemical faecal occult blood test (iFOBT) screening for people aged 50-74 years. Previous work has quantified the number of colorectal cancer (CRC) deaths prevented by the NBCSP and has shown that it is cost-effective. With a 40% screening participation rate, the NBCSP is currently underutilised and could be improved by increasing program participation, but the maximum appropriate level of spending on effective interventions to increase adherence has not yet been quantified.
Objectives: To estimate (i) reductions in CRC cases and deaths for 2020-2040 attributable to, and (ii) the threshold for cost-effective investment (TCEI) in, effective future interventions to improve participation in the NBCSP.
Methods: A comprehensive microsimulation model, Policy1-Bowel, was used to simulate CRC natural history and screening in Australia, considering currently reported NBCSP adherence rates, i.e. iFOBT participation (∼40%) and diagnostic colonoscopy assessment rates (∼70%). Australian residents aged 40-74 were modelled. We evaluated three scenarios: (1) diagnostic colonoscopy assessment increasing to 90%; (2) iFOBT screening participation increasing to 60% by 2020, 70% by 2030 with diagnostic assessment rates of 90%; and (3) iFOBT screening increasing to 90% by 2020 with diagnostic assessment rates of 90%. In each scenario, we estimated CRC incidence and mortality, colonoscopies, costs, and TCEI given indicative willingness-to-pay thresholds of AUD$10,000-$30,000/LYS.
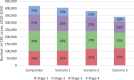
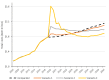
Results: By 2040, age-standardised CRC incidence and mortality rates could be reduced from 46.2 and 13.5 per 100,000 persons, respectively, if current participation rates continued, to (1) 44.0 and 12.7, (2) 36.8 and 8.8, and (3) 31.9 and 6.5. In Scenario 2, 23,000 lives would be saved from 2020-2040 vs current participation rates. The estimated scenario-specific TCEI (Australian dollars or AUD$/year) to invest in interventions to increase participation, given a conservative willingness-to-pay threshold of AUD$10,000/LYS, was (1) AUD$14.9M, (2) AUD$72.0M, and (3) AUD$76.5M.
Conclusion: Significant investment in evidence-based interventions could be used to improve NBCSP adherence and help realise the program's potential. Such interventions might include mass media campaigns to increase program participation, educational or awareness interventions for practitioners, and/or interventions resulting in improvements in referral pathways. Any set of interventions which achieves at least 70% iFOBT screening participation and a 90% diagnostic assessment rate while costing under AUD$72 million annually would be highly cost-effective (<AUD$10,000/LYS) and save 23,000 additional lives from 2020-2040.
Conflict of interest statement
KC is co-PI of an unrelated investigator-initiated trial of cervical screening in Australia (‘Compass’) conducted by the VCS Foundation (a government-funded not-for profit agency). The VCS Foundation has received a funding contribution for Compass from Roche Molecular Systems and Ventana Inc., USA. This does not alter our adherence to PLOS ONE policies on sharing data and materials. The other authors declare no direct conflict of interest.
Figures

References
-
- Welfare AIoHa. Cancer in Australia 2019. Cat. no. CAN 123. Canberra: AIHW; 2019.
-
- von Karsa L, Patnick J, Segnan N. European guidelines for quality assurance in colorectal cancer screening and diagnosis. First Edition—Executive summary. Endoscopy. 2012;44 Suppl 3. - PubMed
-
- Salkeld G, Young G, Irwig L, Haas M, Glasziou P. Cost-effectiveness analysis of screening by faecal occult blood testing for colorectal cancer in Australia. Aust N Z J Public Health. 1996;20(2). - PubMed
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources
Medical

