A Simple in Syringe Low Density Solvent-Dispersive Liquid Liquid Microextraction for Enrichment of Some Metal Ions Prior to Their Determination by High Performance Liquid Chromatography in Food Samples
- PMID: 32012808
- PMCID: PMC7037012
- DOI: 10.3390/molecules25030552
A Simple in Syringe Low Density Solvent-Dispersive Liquid Liquid Microextraction for Enrichment of Some Metal Ions Prior to Their Determination by High Performance Liquid Chromatography in Food Samples
Abstract
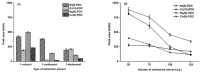
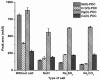
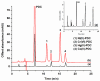
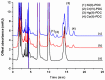
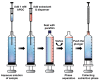
A simple and highly sensitive method is developed for the simultaneous determination of Ni2+, Cr2O72-, Co2+, and Hg2+ by using in syringe low density solvent-dispersive liquid liquid microextraction (ISLD-DLLME), followed by high performance liquid chromatography with a UV detector. The four metal ions were derivatized with pyrrolidine dithiocarbamate (PDC) based on complexation before their enrichment by ISLD-DLLME in which 1-octanol and methanol were used as the extraction solvent and the dispersive solvent, respectively. The extraction was performed in a commercially available syringe under vortex agitation. Phase separation was achieved without centrifugation, and the extraction phase was easily collected by moving the syringe plunger. Parameters affecting the extraction efficiency were studied and optimized. Under the optimum conditions, the four metal-PDC complexes were detected within 18 min, and ISLD-DLLME could increase the detection sensitivity in the range of 64-230 times compared to the direct HPLC analysis. The obtained limits of detection (LODs) were found to be in the range of 0.011-2.0 µg L-1. The applicability of the method is demonstrated for freshwater fish, shrimp, and shellfish samples. In addition, the results are in good agreement with those obtained by inductively-coupled plasma-optical emission spectrometry (ICP-OES).
Keywords: in-syringe microextraction; liquid phase microextraction; metal complex; pyrrolidine dithiocarbamate; simultaneous analysis.
Conflict of interest statement
The authors declare no conflicts of interest.
Figures

Similar articles
-
Low-density solvent-based vortex-assisted surfactant-enhanced-emulsification liquid-liquid microextraction combined with gas chromatography-mass spectrometry for the fast determination of phthalate esters in bottled water.J Chromatogr A. 2013 Jan 25;1274:28-35. doi: 10.1016/j.chroma.2012.12.017. Epub 2012 Dec 19. J Chromatogr A. 2013. PMID: 23290358
-
Application of dispersive liquid-liquid microextraction coupled with vortex-assisted hydrophobic magnetic nanoparticles based solid-phase extraction for determination of aflatoxin M1 in milk samples by sensitive micelle enhanced spectrofluorimetry.Talanta. 2015 Mar;134:98-104. doi: 10.1016/j.talanta.2014.11.007. Epub 2014 Nov 11. Talanta. 2015. PMID: 25618645
-
A simple solvent based microextraction for high performance liquid chromatographic analysis of aflatoxins in rice samples.Talanta. 2018 Jan 1;176:172-177. doi: 10.1016/j.talanta.2017.08.028. Epub 2017 Aug 9. Talanta. 2018. PMID: 28917738
-
Green aspects, developments and perspectives of liquid phase microextraction techniques.Talanta. 2014 Feb;119:34-45. doi: 10.1016/j.talanta.2013.10.050. Epub 2013 Oct 29. Talanta. 2014. PMID: 24401382 Review.
-
Beyond dispersive liquid-liquid microextraction.J Chromatogr A. 2014 Mar 28;1335:2-14. doi: 10.1016/j.chroma.2014.02.021. Epub 2014 Feb 15. J Chromatogr A. 2014. PMID: 24582396 Review.
Cited by
-
Biological Monitoring of Glyphosate Exposure among Knapsack Sprayers in Khon Kaen, Thailand.Toxics. 2024 May 6;12(5):337. doi: 10.3390/toxics12050337. Toxics. 2024. PMID: 38787116 Free PMC article.
-
The Versatility in the Applications of Dithiocarbamates.Int J Mol Sci. 2022 Jan 24;23(3):1317. doi: 10.3390/ijms23031317. Int J Mol Sci. 2022. PMID: 35163241 Free PMC article. Review.
-
Air-assisted cloud point extraction coupled with inductively coupled plasma optical emission spectroscopy for determination of samarium in environmental samples.Anal Sci. 2022 Dec;38(12):1489-1495. doi: 10.1007/s44211-022-00181-9. Epub 2022 Aug 31. Anal Sci. 2022. PMID: 36044121 Free PMC article.
References
-
- Shrivas K., Dewangan K., Ahmed A. Surfactant-based dispersive liquid–liquid microextraction for the determination of zinc in environmental water samples using flame atomic absorption spectrometry. Anal. Methods. 2016;8:5519–5525. doi: 10.1039/C6AY01277A. - DOI
-
- Sakanupongkul A., Sananmuang R., Udnan Y., Ampiah-Bonney R.J., Chaiyasith W.C. Speciation of mercury in water and freshwater fish samples by a two-step solidified floating organic drop microextraction with electrothermal atomic absorption spectrometry. Food Chem. 2019;277:496–503. doi: 10.1016/j.foodchem.2018.10.131. - DOI - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical

