Anti-MAdCAM-1 antibody (PF-00547659) for active refractory Crohn's disease in Japanese and Korean patients: the OPERA study
- PMID: 32013314
- PMCID: PMC7000638
- DOI: 10.5217/ir.2019.00039
Anti-MAdCAM-1 antibody (PF-00547659) for active refractory Crohn's disease in Japanese and Korean patients: the OPERA study
Abstract
Background/aims: PF-00547659 is a monoclonal antibody against human mucosal addressin cell adhesion molecule-1 (MAdCAM-1) that prevents the binding of α4β7+ lymphocytes to MAdCAM-expressing sites in the gastrointestinal tract with high affinity and selectivity, and is being developed for the treatment of Crohn's disease (CD).
Methods: OPERA is a randomized, multicenter, double-blind, placebo-controlled study to investigate the efficacy, safety, and pharmacokinetics of PF-00547659 following subcutaneous administration in subjects with active CD, a history of failure or intolerance to anti-tumor necrosis factor and/or immunosuppressants, high-sensitivity C-reactive protein > 3.0 mg/L, and ulcers on colonoscopy. The primary endpoint was Crohn's Disease Activity Index-70 response at week 8 or 12. Subpopulation analyses for Asian subjects were performed as some differences are observed in genetics and clinical phenotypes in Asian CD patients compared with Western patients.
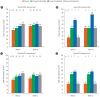
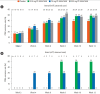
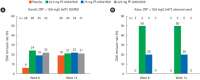
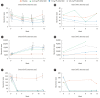
Results: In this study, 265 CD subjects were randomized, with a subpopulation of 21 subjects (8 Japanese and 13 Korean) defined as the Asian population. In the overall and Asian populations; PF-00547659 was pharmacologically active as evidenced by soluble MAdCAM and circulating β7+ central memory CD4+ T-lymphocytes, although no clear evidence of efficacy was observed in any clinical endpoints; pharmacokinetics of PF-00547659 in the Asian subpopulation was generally comparable to the overall population; and the safety profile of PF-00547659 appeared acceptable up to 12 weeks of treatment.
Conclusions: In the overall and Asian populations, efficacy of PF-00547659 could not be demonstrated using any clinical endpoints compared with placebo. Pharmacokinetics and safety of PF-00547659 were generally comparable. Further studies with larger numbers of patients are required to confirm our results. (Trial Registration Number: NCT01276509).
Keywords: Crohn disease; Japanese; Korean; MAdCAM; PF-00547659.
Conflict of interest statement
This study was sponsored by Pfizer. Banerjee A, Isogawa N, Li Y, and Hassan-Zahraee M are employees of Pfizer. Ahmad A, Cataldi F, Gorelick KJ, Nagaoka M, and Clare R were employees of Pfizer during the OPERA study. Saruta M received lecture fees from Mitsubishi Tanabe Pharma, AbbVie GK, and Takeda Pharmaceutical Co., Ltd., and research grants from EA Pharma Co., Ltd., Kissei Pharmaceutical Co., Ltd., Otsuka Pharmaceutical Co., Ltd., and Mochida Pharmaceutical Co., Ltd. Watanabe M has the following disclosures: honoraria from Mitsubishi Tanabe Pharma Corp., Eisai Co., Ltd., Kyorin Pharmaceutical Co., Ltd., JIMRO Co., Ltd., AbbVie GK, Takeda Pharmaceutical Co., Ltd., Kyowa Hakko Kirin Co., Ltd., Zeria Pharmaceutical Co., Ltd., Asahi Kasei Medical Co., Ltd., EA Pharma Co., Ltd., Astellas Pharma Inc., Mochida Pharmaceutical Co., Ltd., Janssen Pharmaceutical Co., Ltd., Gilead Sciences, Inc., Celgene Corp., KISSEI Pharmaceutical Co., Ltd., and commercial research funding from: Asahi Kasei Medical Co., Ltd., AbbVie GK, EA Pharma Co., Ltd., Eisai Co., Ltd., Kyorin Phar maceutical Co., Ltd., Mitsubishi Tanabe Pharma Corp., Otsuka Pharmaceutical Co., Ltd., Kyowa Hakko Kirin Co., Ltd., Zeria Pharmaceutical Co., Ltd., JIMRO Co., Ltd., Takeda Pharmaceutical Co., Ltd., Nippon Kayaku Co., Ltd., Mochida Pharmaceutical Co., Ltd., Daiichi Sankyo Co., Ltd., Astellas Pharma Inc., MSD K.K., Dainippon Sumitomo Dainippon Pharma Co., Ltd., Bristol-Myers, K.K, Chugai Pharmaceutical Co., Ltd., Gilead Sciences, Inc., Pfizer Inc., Miyarisan Pharmaceutical Co., Ltd., KISSEI Pharmaceutical Co., Ltd., Taiho Pharmaceutical Co., Ltd. Jang BI, Park DI, Cheon JH, Im JP, Yang SK, Hibi T, Kanai T, Katsuno T, Kim YH, and Li Y have no disclosures. However, all of these are not relevant to this article.
Figures
Similar articles
-
Phase II evaluation of anti-MAdCAM antibody PF-00547659 in the treatment of Crohn's disease: report of the OPERA study.Gut. 2018 Oct;67(10):1824-1835. doi: 10.1136/gutjnl-2016-313457. Epub 2017 Oct 5. Gut. 2018. PMID: 28982740 Free PMC article. Clinical Trial.
-
Anti-MAdCAM Antibody Increases ß7+ T Cells and CCR9 Gene Expression in the Peripheral Blood of Patients With Crohn's Disease.J Crohns Colitis. 2018 Jan 5;12(1):77-86. doi: 10.1093/ecco-jcc/jjx121. J Crohns Colitis. 2018. PMID: 28961803 Free PMC article. Clinical Trial.
-
Effect of PF-00547659 on Central Nervous System Immune Surveillance and Circulating β7+ T Cells in Crohn's Disease: Report of the TOSCA Study.J Crohns Colitis. 2018 Jan 24;12(2):188-196. doi: 10.1093/ecco-jcc/jjx128. J Crohns Colitis. 2018. PMID: 28961770 Free PMC article.
-
PF-00547659 for the treatment of Crohn's disease and ulcerative colitis.Expert Opin Investig Drugs. 2018 Jul;27(7):623-629. doi: 10.1080/13543784.2018.1494722. Epub 2018 Jul 16. Expert Opin Investig Drugs. 2018. PMID: 29985060 Review.
-
Certolizumab pegol for the management of Crohn's disease in adults.Clin Ther. 2009 Jun;31(6):1158-76. doi: 10.1016/j.clinthera.2009.06.015. Clin Ther. 2009. PMID: 19695385 Review.
Cited by
-
Targeting Immune Cell Trafficking - Insights From Research Models and Implications for Future IBD Therapy.Front Immunol. 2021 May 4;12:656452. doi: 10.3389/fimmu.2021.656452. eCollection 2021. Front Immunol. 2021. PMID: 34017333 Free PMC article. Review.
-
Novel Histone Deacetylase 6 Inhibitor Confers Anti-inflammatory Effects and Enhances Gut Barrier Function.Gut Liver. 2023 Sep 15;17(5):766-776. doi: 10.5009/gnl220159. Epub 2022 Sep 27. Gut Liver. 2023. PMID: 36167345 Free PMC article.
-
Emerging therapeutic options in inflammatory bowel disease.World J Gastroenterol. 2021 Dec 28;27(48):8242-8261. doi: 10.3748/wjg.v27.i48.8242. World J Gastroenterol. 2021. PMID: 35068868 Free PMC article. Review.
-
Downregulation of Heat Shock Protein 72 Contributes to Fibrostenosis in Crohn's Disease.Gut Liver. 2023 Nov 15;17(6):905-915. doi: 10.5009/gnl220308. Epub 2023 Feb 23. Gut Liver. 2023. PMID: 36814356 Free PMC article.
References
-
- Fiocchi C. Inflammatory bowel disease pathogenesis: where are we? J Gastroenterol Hepatol. 2015;30 Suppl 1:12–18. - PubMed
-
- Streeter PR, Berg EL, Rouse BT, Bargatze RF, Butcher EC. A tissue-specific endothelial cell molecule involved in lymphocyte homing. Nature. 1988;331:41–46. - PubMed
-
- Nakache M, Berg EL, Streeter PR, Butcher EC. The mucosal vascular addressin is a tissue-specific endothelial cell adhesion molecule for circulating lymphocytes. Nature. 1989;337:179–181. - PubMed
Associated data
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Research Materials

