Dissociated Hippocampal Neurons Exhibit Distinct Zn2+ Dynamics in a Stimulation-Method-Dependent Manner
- PMID: 32013397
- PMCID: PMC7251562
- DOI: 10.1021/acschemneuro.0c00006
Dissociated Hippocampal Neurons Exhibit Distinct Zn2+ Dynamics in a Stimulation-Method-Dependent Manner
Abstract
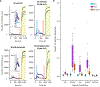
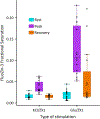
Ionic Zn2+ has increasingly been recognized as an important neurotransmitter and signaling ion in glutamatergic neuron pathways. Intracellular Zn2+ transiently increases as a result of neuronal excitation, and this Zn2+ signal is essential for neuron plasticity, but the source and regulation of the signal is still unclear. In this study, we rigorously quantified Zn2+, Ca2+, and pH dynamics in dissociated mouse hippocampal neurons stimulated with bath application of high KCl or glutamate. While both stimulation methods yielded Zn2+ signals, Ca2+ influx, and acidification, glutamate stimulation induced more sustained high intracellular Ca2+ and a larger increase in intracellular Zn2+. However, the stimulation-induced pH change was similar between conditions, indicating that a different cellular change is responsible for the stimulation-dependent difference in Zn2+ signal. This work provides the first robust quantification of Zn2+ dynamics in neurons using different methods of stimulation.
Keywords: Zinc imaging; calcium imaging; hippocampal culture; neurotransmitters; pH imaging; signaling.
Conflict of interest statement
The authors declare no competing financial interest.
Figures


References
-
- Andreini C; Banci L; Bertini I; Rosato A Counting the Zinc-Proteins Encoded in the Human Genome. J. Proteome Res 2006, 5 (1), 196–201. - PubMed
-
- Levaot N; Hershfinkel M How Cellular Zn2+ Signaling Drives Physiological Functions. Cell Calcium 2018, 75, 53–63. - PubMed
-
- Claiborne BJ; Rea MA; Terrian DM Detection of Zinc in Isolated Nerve Terminals Using a Modified Timm’s Sulfide-Silver Method. J. Neurosci. Methods 1989, 30 (1), 17–22. - PubMed
-
- Frederickson CJ; Danscher G Zinc-Containing Neurons in Hippocampus and Related CNS Structures. Prog. Brain Res 1990, 83, 71–84. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous

