Twist-1 is upregulated by NSD2 and contributes to tumour dissemination and an epithelial-mesenchymal transition-like gene expression signature in t(4;14)-positive multiple myeloma
- PMID: 32014459
- PMCID: PMC7829063
- DOI: 10.1016/j.canlet.2020.01.040
Twist-1 is upregulated by NSD2 and contributes to tumour dissemination and an epithelial-mesenchymal transition-like gene expression signature in t(4;14)-positive multiple myeloma
Abstract
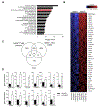
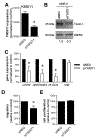
Approximately 15% of patients with multiple myeloma (MM) harbour the t(4;14) chromosomal translocation, leading to the overexpression of the histone methyltransferase NSD2. Patients with this translocation display increased tumour dissemination, accelerated disease progression and rapid relapse. Using publicly available gene expression profile data from NSD2high (n = 135) and NSD2low (n = 878) MM patients, we identified 39 epithelial-mesenchymal transition (EMT)-associated genes which are overexpressed in NSD2high MM plasma cells. In addition, our analyses identified Twist-1 as a key transcription factor upregulated in NSD2high MM patients and t(4;14)-positive cell lines. Overexpression and knockdown studies confirmed that Twist-1 is involved in driving the expression of EMT-associated genes in the human MM cell line KMS11 and promoted the migration of myeloma cell lines in vitro. Notably, Twist-1 overexpression in the mouse MM cell line 5TGM1 significantly increased tumour dissemination in an intratibial tumour model. These findings demonstrate that Twist-1, downstream of NSD2, contributes to the induction of an EMT-like signature in t(4;14)-positive MM and enhances the dissemination of MM plasma cells in vivo, which may, in part, explain the aggressive disease features associated with t(4;14)-positive MM.
Keywords: Dissemination; Epithelial-mesenchymal transition; Multiple myeloma; NSD2; Twist-1.
Copyright © 2020 Elsevier B.V. All rights reserved.
Conflict of interest statement
Declaration of competing interest All authors declare no competing financial interests.
Figures

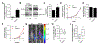
References
-
- Dutta AK, Hewett DR, Fink JL, Grady JP, Zannettino ACW, Cutting edge genomics reveal new insights into tumour development, disease progression and therapeutic impacts in multiple myeloma, Br. J. Haematol, (2017). - PubMed
-
- Rawstron AC, Owen RG, Davies FE, Johnson RJ, Jones RA, Richards SJ, Evans PA, Child JA, Smith GM, Jack AS, Morgan GJ, Circulating plasma cells in multiple myeloma: characterization and correlation with disease stage, Br J Haematol, 97 (1997) 46–55. - PubMed
-
- Bergsagel PL, Kuehl WM, Chromosome translocations in multiple myeloma, Oncogene, 20 (2001) 5611–5622. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical

