Efficacy of Early Oral Switch with β-Lactams for Low-Risk Staphylococcus aureus Bacteremia
- PMID: 32015029
- PMCID: PMC7318028
- DOI: 10.1128/AAC.02345-19
Efficacy of Early Oral Switch with β-Lactams for Low-Risk Staphylococcus aureus Bacteremia
Abstract
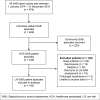
The aim of this study was to assess the safety of early oral switch (EOS) prior to 14 days for low-risk Staphylococcus aureus bacteremia (LR-SAB), which is the primary treatment strategy used at our institution. The usual recommended therapy is 14 days of intravenous (i.v.) antibiotics. All patients with SAB at our hospital were identified between 1 January 2014 and 31 December 2018. Those meeting low-risk criteria (health care-associated, no evidence of deep infection or demonstrated involvement of prosthetic material, and no further positive blood cultures after 72 h) were included in the study. The primary outcome was occurrence of a SAB-related complication within 90 days. There were 469 SAB episodes during the study period, 100 (21%) of whom met inclusion criteria. EOS was performed in 84 patients. In this group, line infection was the source in 79%, methicillin-susceptible S. aureus caused 95% of SABs and 74% of patients received i.v. flucloxacillin. The median durations of i.v. and oral antibiotics in the EOS group were 5 days (interquartile range [IQR], 4 to 6) and 10 days (IQR, 9 to 14), respectively. A total of 71% of patients received flucloxacillin as their EOS agent. Overall, 86% of oral step-down therapy was with beta-lactams. One patient (1%) undergoing EOS had SAB relapse within 90 days. No deaths attributable to SAB occurred within 90 days. In this low-MRSA-prevalence LR-SAB cohort, EOS was associated with a low incidence of SAB-related complications. This was achieved with oral beta-lactam therapy in most patients. Larger prospective studies are needed to confirm these findings.
Keywords: Staphylococcus aureus bacteremia; beta-lactam; early oral switch; low risk.
Copyright © 2020 American Society for Microbiology.
Figures
Comment in
-
Early Oral Antibiotic Switch for Staphylococcus aureus Bacteremia: Many Are Called, but Few Are Chosen.Antimicrob Agents Chemother. 2020 Jun 23;64(7):e00317-20. doi: 10.1128/AAC.00317-20. Print 2020 Jun 23. Antimicrob Agents Chemother. 2020. PMID: 32393495 Free PMC article.
References
-
- Liu C, Infectious Diseases Society of America, Bayer A, Cosgrove SE, Daum RS, Fridkin SK, Gorwitz RJ, Kaplan SL, Karchmer AW, Levine DP, Murray BE, Rybak MJ, Talan DA, Chambers HF. 2011. Clinical practice guidelines by the Infectious Diseases Society of America for the treatment of methicillin-resistant Staphylococcus aureus infections in adults and children. Clin Infect Dis 52:e18–e55. doi: 10.1093/cid/ciq146. - DOI - PubMed
-
- Thwaites GE, UK Clinical Infection Research Group, Edgeworth JD, Gkrania-Klotsas E, Kirby A, Tilley R, Török ME, Walker S, Wertheim HF, Wilson P, Llewelyn MJ. 2011. Clinical management of Staphylococcus aureus bacteraemia. Lancet Infect Dis 11:208–222. doi: 10.1016/S1473-3099(10)70285-1. - DOI - PubMed
-
- Gould FK, Brindle R, Chadwick PR, Fraise AP, Hill S, Nathwani D, Ridgway GL, Spry MJ, Warren RE, MRSA Working Party of the British Society for Antimicrobial Chemotherapy . 2009. Guidelines (2008) for the prophylaxis and treatment of methicillin-resistant Staphylococcus aureus (MRSA) infections in the United Kingdom. J Antimicrob Chemother 63:849–861. doi: 10.1093/jac/dkp065. - DOI - PubMed
-
- Mermel LA, Allon M, Bouza E, Craven DE, Flynn P, O’Grady NP, Raad II, Rijnders BJA, Sherertz RJ, Warren DK. 2009. Clinical practice guidelines for the diagnosis and management of intravascular catheter-related infection: 2009 update by the Infectious Diseases Society of America. Clin Infect Dis 49:1–45. doi: 10.1086/599376. - DOI - PMC - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous