NAD deficiency due to environmental factors or gene-environment interactions causes congenital malformations and miscarriage in mice
- PMID: 32015132
- PMCID: PMC7035598
- DOI: 10.1073/pnas.1916588117
NAD deficiency due to environmental factors or gene-environment interactions causes congenital malformations and miscarriage in mice
Abstract
Causes for miscarriages and congenital malformations can be genetic, environmental, or a combination of both. Genetic variants, hypoxia, malnutrition, or other factors individually may not affect embryo development, however, they may do so collectively. Biallelic loss-of-function variants in HAAO or KYNU, two genes of the nicotinamide adenine dinucleotide (NAD) synthesis pathway, are causative of congenital malformation and miscarriage in humans and mice. The variants affect normal embryonic development by disrupting the synthesis of NAD, a key factor in multiple biological processes, from its dietary precursor tryptophan, resulting in NAD deficiency. This study demonstrates that congenital malformations caused by NAD deficiency can occur independent of genetic disruption of NAD biosynthesis. C57BL/6J wild-type mice had offspring exhibiting similar malformations when their supply of the NAD precursors tryptophan and vitamin B3 in the diet was restricted during pregnancy. When the dietary undersupply was combined with a maternal heterozygous variant in Haao, which alone does not cause NAD deficiency or malformations, the incidence of embryo loss and malformations was significantly higher, suggesting a gene-environment interaction. Maternal and embryonic NAD levels were deficient. Mild hypoxia as an additional factor exacerbated the embryo outcome. Our data show that NAD deficiency as a cause of embryo loss and congenital malformation is not restricted to the rare cases of biallelic mutations in NAD synthesis pathway genes. Instead, monoallelic genetic variants and environmental factors can result in similar outcomes. The results expand our understanding of the causes of congenital malformations and the importance of sufficient NAD precursor consumption during pregnancy.
Keywords: NAD; congenital malformation; embryonic development; metabolism; miscarriage.
Conflict of interest statement
The authors declare no competing interest.
Figures


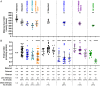
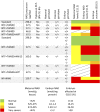
Comment in
-
Dietary modification, penetrance, and the origins of congenital malformation.Proc Natl Acad Sci U S A. 2020 Mar 10;117(10):5097-5099. doi: 10.1073/pnas.2000944117. Epub 2020 Feb 18. Proc Natl Acad Sci U S A. 2020. PMID: 32071229 Free PMC article. No abstract available.
References
-
- Rai R., Regan L., Recurrent miscarriage. Lancet 368, 601–611 (2006). - PubMed
-
- Kaiser J., Branch D. W., Recurrent pregnancy loss: Generally accepted causes and their management. Clin. Obstet. Gynecol. 59, 464–473 (2016). - PubMed
-
- Baldacci S., et al. , Environmental and individual exposure and the risk of congenital anomalies: A review of recent epidemiological evidence. Epidemiol. Prev. 42 (suppl. 1), 1–34 (2018). - PubMed
-
- Zhu H., Kartiko S., Finnell R. H., Importance of gene-environment interactions in the etiology of selected birth defects. Clin. Genet. 75, 409–423 (2009). - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Molecular Biology Databases

