Truly continuous low pH viral inactivation for biopharmaceutical process integration
- PMID: 32017010
- PMCID: PMC7187162
- DOI: 10.1002/bit.27292
Truly continuous low pH viral inactivation for biopharmaceutical process integration
Abstract
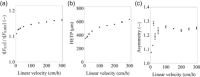
Continuous virus inactivation (VI) has received little attention in the efforts to realize fully continuous biomanufacturing in the future. Implementation of continuous VI must assure a specific minimum incubation time, typically 60 min. To guarantee the minimum incubation time, we implemented a packed bed continuous viral inactivation reactor (CVIR) with narrow residence time distribution (RTD) for low pH incubation. We show that the RTD does not broaden significantly over a wide range of linear flow velocities-which highlights the flexibility and robustness of the design. Prolonged exposure to acidic pH has no impact on bed stability, assuring constant RTD throughout long term operation. The suitability of the packed bed CVIR for low pH inactivation is shown with two industry-standard model viruses, that is xenotropic murine leukemia virus and pseudorabies virus. Controls at neutral pH showed no system-induced VI. At low pH, significant VI is observed, even after only 15 min. Based on the low pH inactivation kinetics, the continuous process is equivalent to traditional batch operation. This study establishes a concept for continuous low pH inactivation and, together with previous reports, highlights the versatility of the packed bed reactor for continuous VI, regardless of the inactivation method.
Keywords: continuous processing; low pH viral inactivation; residence time distribution; virus clearance; virus inactivation.
© 2020 The Authors. Biotechnology and Bioengineering Published by Wiley Periodicals, Inc.
Conflict of interest statement
The authors declare that there are no conflict of interests.
Figures





References
-
- Albeladi, H. K. , Al‐Romaizan, A. N. , & Hussein, M. A. (2017). Role of cross‐linking process on the performance of PMMA. International Journal of Biosensors & Bioelectronics, 3(3), 279–284. 10.15406/ijbsbe.2017.03.00065 - DOI
-
- Ali, U. , Bt. Abd Karim, K. J. , & Buang, N. A. (2015). A review of the properties and applications of poly (methyl methacrylate) (PMMA). Polymer Reviews, 55(4), 678–705. 10.1080/15583724.2015.1031377 - DOI
-
- Andersson Trojer, M. , Wendel, A. , Holmberg, K. , & Nydén, M. (2012). The effect of pH on charge, swelling and desorption of the dispersant poly(methacrylic acid) from poly(methyl methacrylate) microcapsules. Journal of Colloid and Interface Science, 375(1), 213–215. 10.1016/J.JCIS.2012.02.026 - DOI - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

