Sustained treatment of retinal vascular diseases with self-aggregating sunitinib microparticles
- PMID: 32019921
- PMCID: PMC7000758
- DOI: 10.1038/s41467-020-14340-x
Sustained treatment of retinal vascular diseases with self-aggregating sunitinib microparticles
Abstract
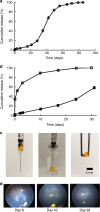
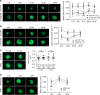
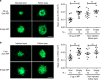
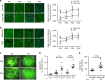
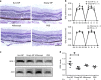
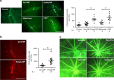
Neovascular age-related macular degeneration and diabetic retinopathy are prevalent causes of vision loss requiring frequent intravitreous injections of VEGF-neutralizing proteins, and under-treatment is common and problematic. Here we report incorporation of sunitinib, a tyrosine kinase inhibitor that blocks VEGF receptors, into a non-inflammatory biodegradable polymer to generate sunitinib microparticles specially formulated to self-aggregate into a depot. A single intravitreous injection of sunitinib microparticles potently suppresses choroidal neovascularization in mice for six months and in another model, blocks VEGF-induced leukostasis and retinal nonperfusion, which are associated with diabetic retinopathy progression. After intravitreous injection in rabbits, sunitinib microparticles self-aggregate into a depot that remains localized and maintains therapeutic levels of sunitinib in retinal pigmented epithelium/choroid and retina for more than six months. There is no intraocular inflammation or retinal toxicity. Intravitreous injection of sunitinib microparticles provides a promising approach to achieve sustained suppression of VEGF signaling and improve outcomes in patients with retinal vascular diseases.
Conflict of interest statement
J.H., J.F. and P.A.C. are founders of Graybug Vision which has a commercial interest in sunitinib microparticles, and they have equity in Graybug and receive remuneration for consultation. Johns Hopkins University has patents on which J.H. and J.F. are authors that are licensed to Graybug Vision. All of these relationships are being managed by the Johns Hopkins University conflict of interest committee. Y.Y., D.M., D.Ca., W.Y. and M.Y. are employees of Graybug Vision. J.K., W.P., J.Z.Z. and J.L.C. are former employees of Graybug Vision. The remaining authors declare no competing interests.
Figures

References
-
- Kwak N, Okamoto N, Wood JM, Campochiaro PA. VEGF is an important stimulator in a model of choroidal neovascularization. Invest. Ophthalmol. Vis. Sci. 2000;41:3158–3164. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases

