The Added Value of Diagnostic and Theranostic PET Imaging for the Treatment of CNS Tumors
- PMID: 32033160
- PMCID: PMC7037158
- DOI: 10.3390/ijms21031029
The Added Value of Diagnostic and Theranostic PET Imaging for the Treatment of CNS Tumors
Abstract
This review highlights the added value of PET imaging in Central Nervous System (CNS) tumors, which is a tool that has rapidly evolved from a merely diagnostic setting to multimodal molecular diagnostics and the guidance of targeted therapy. PET is the method of choice for studying target expression and target binding behind the assumedly intact blood-brain barrier. Today, a variety of diagnostic PET tracers can be used for the primary staging of CNS tumors and to determine the effect of therapy. Additionally, theranostic PET tracers are increasingly used in the context of pharmaceutical and radiopharmaceutical drug development and application. In this approach, a single targeted drug is used for PET diagnosis, upon the coupling of a PET radionuclide, as well as for targeted (nuclide) therapy. Theranostic PET tracers have the potential to serve as a non-invasive whole body navigator in the selection of the most effective drug candidates and their most optimal dose and administration route, together with the potential to serve as a predictive biomarker in the selection of patients who are most likely to benefit from treatment. PET imaging supports the transition from trial and error medicine to predictive, preventive, and personalized medicine, hopefully leading to improved quality of life for patients and more cost-effective care.
Keywords: CNS tumors; PET; central nervous system; drug development; molecular biology; molecular imaging; oncology; positron emission tomography; targeted therapy; theranostics.
Conflict of interest statement
The authors declare no conflict of interest.
Figures
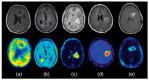
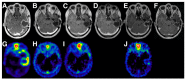
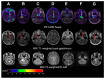
References
-
- Food and Drug Administration (FDA) In vitro companion diagnostic devices: Guidance for industry and Food and Drug Administration staff. [(accessed on 3 January 2020)]; Available online: https://www.fda.gov/downloads/MedicalDevices/DeviceRegulationandGuidance....
-
- Food and Drug Administration (FDA) List of cleared or approved companion diagnostic devices (in vitro and imaging tools) [(accessed on 3 January 2020)]; Available online: https://www.fda.gov/medical-devices/vitro-diagnostics/list-cleared-or-ap....
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources

