Permanent Photodynamic Activation of the Cholecystokinin 2 Receptor
- PMID: 32033232
- PMCID: PMC7072308
- DOI: 10.3390/biom10020236
Permanent Photodynamic Activation of the Cholecystokinin 2 Receptor
Abstract
The cholecystokinin 2 receptor (CCK2R) is expressed in the central nervous system and peripheral tissues, playing an important role in higher nervous and gastrointestinal functions, pain sensation, and cancer growth. CCK2R is reversibly activated by cholecystokinin or gastrin, but whether it can be activated permanently is not known. In this work, we found that CCK2R expressed ectopically in CHO-K1 cells was permanently activated in the dark by sulfonated aluminum phthalocyanine (SALPC / AlPcS4, 10-1,000 nM), as monitored by Fura-2 fluorescent calcium imaging. Permanent CCK2R activation was also observed with AlPcS2, but not PcS4. CCK2R previously exposed to SALPC (3 and 10 nM) was sensitized by subsequent light irradiation (> 580 nm, 31.5 mW·cm-2). After the genetically encoded protein photosensitizer mini singlet oxygen generator (miniSOG) was fused to the N-terminus of CCK2R and expressed in CHO-K1 cells, light irradiation (450 nm, 85 mW·cm-2) activated in-frame CCK2R (miniSOG-CCK2R), permanently triggering persistent calcium oscillations blocked by the CCK2R antagonist YM 022 (30 nM). From these data, it is concluded that SALPC is a long-lasting CCK2R agonist in the dark, and CCK2R is photogenetically activated permanently with miniSOG as photosensitizer. These properties of SALPC and CCK2R could be used to study CCK2R physiology and possibly for pain and cancer therapies.
Keywords: CCK2R; SALPC; calcium oscillations; miniSOG; photodynamic action.
Conflict of interest statement
The authors declare no conflicts of interest. The funders had no role in the design of the study; in the collection, analyses, or interpretation of data; in the writing of the manuscript, or in the decision to publish the results.
Figures
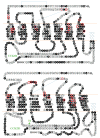


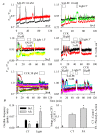

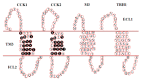
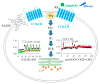
References
-
- Tang N., Zhang X., Wang S., Qi J., Tian Z., Wang B., Chen H., Wu Y., Wang M., Xu S., et al. UCN3 suppresses food intake in coordination with CCK and the CCK2R in Siberian sturgeon (Acipenser baerii) Comp. Biochem. Physiol. Part A Mol. Integr. Physiol. 2019;234:106–113. doi: 10.1016/j.cbpa.2019.04.022. - DOI - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

