Efficacy, Safety, and Acceptability of Pharmacologic Treatments for Pediatric Migraine Prophylaxis: A Systematic Review and Network Meta-analysis
- PMID: 32040139
- PMCID: PMC7042942
- DOI: 10.1001/jamapediatrics.2019.5856
Efficacy, Safety, and Acceptability of Pharmacologic Treatments for Pediatric Migraine Prophylaxis: A Systematic Review and Network Meta-analysis
Abstract
Importance: Migraine is one of the most common neurologic disorders in children and adolescents. However, a quantitative comparison of multiple preventive pharmacologic treatments in the pediatric population is lacking.
Objective: To examine whether prophylactic pharmacologic treatments are more effective than placebo and whether there are differences between drugs regarding efficacy, safety, and acceptability.
Data sources: Systematic review and network meta-analysis of studies in MEDLINE, Cochrane, Embase, and PsycINFO published through July 2, 2018.
Study selection: Randomized clinical trials of prophylactic pharmacologic treatments in children and adolescents diagnosed as having episodic migraine were included. Abstract, title, and full-text screening were conducted independently by 4 reviewers.
Data extraction and synthesis: Data extraction was conducted according to Preferred Reporting Items for Systematic Reviews and Meta-Analysis network meta-analysis guidelines. Quality was assessed with the Cochrane Risk of Bias tool. Effect sizes, calculated as standardized mean differences for primary outcomes and risk ratios for discontinuation rates, were assessed in a random-effects model.
Main outcomes and measures: Primary outcomes were efficacy (ie, migraine frequency, number of migraine days, number of headache days, headache frequency, or headache index), safety (ie, treatment discontinuation owing to adverse events), and acceptability (ie, treatment discontinuation for any reason).
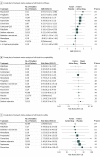
Results: Twenty-three studies (2217 patients) were eligible for inclusion. Prophylactic pharmacologic treatments included antiepileptics, antidepressants, calcium channel blockers, antihypertensive agents, and food supplements. In the short term (<5 months), propranolol (standard mean difference, 0.60; 95% CI, 0.03-1.17) and topiramate (standard mean difference, 0.59; 95% CI, 0.03-1.15) were significantly more effective than placebo. However, the 95% prediction intervals for these medications contained the null effect. No significant long-term effects for migraine prophylaxis relative to placebo were found for any intervention.
Conclusions and relevance: Prophylactic pharmacologic treatments have little evidence supporting efficacy in pediatric migraine. Future research could (1) identify factors associated with individual responses to pharmacologic prophylaxis, (2) analyze fluctuations of migraine attack frequency over time and determine the most clinically relevant length of probable prophylactic treatment, and (3) identify nonpharmacologic targets for migraine prophylaxis.
Conflict of interest statement
Figures

Comment in
-
Can We Really Stop Pediatric Migraine?: Using Network Meta-analysis to Remove the Guess Work.JAMA Pediatr. 2020 Apr 1;174(4):325-326. doi: 10.1001/jamapediatrics.2019.5907. JAMA Pediatr. 2020. PMID: 32040138 No abstract available.
Similar articles
-
Migraine in Adults: Preventive Pharmacologic Treatments [Internet].Rockville (MD): Agency for Healthcare Research and Quality (US); 2013 Apr. Report No.: 13-EHC068-EF. Rockville (MD): Agency for Healthcare Research and Quality (US); 2013 Apr. Report No.: 13-EHC068-EF. PMID: 23700633 Free Books & Documents. Review.
-
European Headache Federation (EHF) critical re-appraisal and meta-analysis of oral drugs in migraine prevention - part 4: propranolol.J Headache Pain. 2024 Jul 24;25(1):119. doi: 10.1186/s10194-024-01826-y. J Headache Pain. 2024. PMID: 39044170 Free PMC article.
-
Efficacy and safety of cinnarizine in the prophylaxis of migraine headaches in children: an open, randomized comparative trial with propranolol.Acta Neurol Belg. 2012 Mar;112(1):51-5. doi: 10.1007/s13760-012-0011-7. Epub 2012 Jan 24. Acta Neurol Belg. 2012. PMID: 22427290 Clinical Trial.
-
Non-CGRP Antagonist/Non-Triptan Options for Migraine Disease Treatment: Clinical Considerations.Curr Pain Headache Rep. 2023 Oct;27(10):497-502. doi: 10.1007/s11916-023-01151-0. Epub 2023 Aug 16. Curr Pain Headache Rep. 2023. PMID: 37584847 Review.
-
Topiramate as an adjunctive treatment in migraine prophylaxis.Headache. 2003 Nov-Dec;43(10):1080-4. doi: 10.1046/j.1526-4610.2003.03209.x. Headache. 2003. PMID: 14629243 Clinical Trial.
Cited by
-
From the New Diagnostic Criteria to COVID-19 Pandemic Passing Through the Placebo Effect. What Have We Learned in the Management of Pediatric Migrane Over the Past 5 Years?Front Neurol. 2022 Jul 13;13:935803. doi: 10.3389/fneur.2022.935803. eCollection 2022. Front Neurol. 2022. PMID: 35911893 Free PMC article. Review.
-
The use of pharmacologic prophylaxis for migraine in pediatric and adolescent age: a crucial concern.Neurol Sci. 2022 Mar;43(3):2121-2122. doi: 10.1007/s10072-021-05827-6. Epub 2022 Jan 14. Neurol Sci. 2022. PMID: 35028781 No abstract available.
-
Animal-Assisted Psychotherapy for Pediatric Chronic Pain: Case Series of an Open Pilot Study to Test Initial Feasibility and Potential Efficacy.J Pain Res. 2023 May 29;16:1799-1811. doi: 10.2147/JPR.S394270. eCollection 2023. J Pain Res. 2023. PMID: 37273273 Free PMC article.
-
Development and validation of a diagnostic model for migraine without aura in inpatients.Front Neurol. 2025 Jan 21;16:1511252. doi: 10.3389/fneur.2025.1511252. eCollection 2025. Front Neurol. 2025. PMID: 39906147 Free PMC article.
-
Pharmacokinetics and safety of erenumab in pediatric patients with migraine: A phase I, randomized, open-label, multiple-dose study.Clin Transl Sci. 2024 Mar;17(3):e13755. doi: 10.1111/cts.13755. Clin Transl Sci. 2024. PMID: 38476099 Free PMC article. Clinical Trial.
References
-
- National Headache Foundation. FDA approves treximet for adolescents. https://headaches.org/2015/06/26/fda-approves-treximet-for-adolescents/. Accessed June 6, 2019.
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical

