Immunotherapy by Immune Checkpoint Inhibitors and Nuclear Medicine Imaging: Current and Future Applications
- PMID: 32041105
- PMCID: PMC7072145
- DOI: 10.3390/cancers12020371
Immunotherapy by Immune Checkpoint Inhibitors and Nuclear Medicine Imaging: Current and Future Applications
Abstract
Immunotherapy by using immune checkpoint inhibitors is a revolutionary development in oncology. Medical imaging is also impacted by this new therapy, particularly nuclear medicine imaging (also called radionuclide imaging), which uses radioactive tracers to visualize metabolic functions. Our aim was to review the current applications of nuclear medicine imaging in immunotherapy, along with their limitations, and the perspectives offered by this imaging modality. Method: Articles describing the use of radionuclide imaging in immunotherapy were researched using PubMed by April 2019 and analyzed. Results: More than 5000 articles were analyzed, and nearly 100 of them were retained. Radionuclide imaging, notably 18F-FDG PET/CT, already has a major role in many cancers for pre-therapeutic and therapeutic evaluation, diagnoses of adverse effects, called immune-related adverse events (IrAE), and end-of-treatment evaluations. However, these current applications can be hindered by immunotherapy, notably due to atypical response patterns such as pseudoprogression, which is defined as an increase in the size of lesions, or the visualization of new lesions, followed by a response, and hyperprogression, which is an accelerated tumor growth rate after starting treatment. To overcome these difficulties, new opportunities are offered, particularly therapeutic evaluation criteria adapted to immunotherapy and immuno-PET allowing us to predict responses to immunotherapy. Moreover, some new technological solutions are also promising, such as radiomic analyses and body composition on associated anatomical images. However, more research has to be done, notably for the diagnosis of hyperprogression and pseudoprogression. Conclusion: Immunotherapy, by its major impact on cancer and by the new patterns generated on images, is revolutionary in the field of medical images. Nuclear medicine imaging is already established and will be able to help meet new challenges through its plasticity.
Keywords: Adoptive; CD8-Positive T-Lymphocytes; CTLA-4 Antigen; Immunotherapy; diagnostic imaging; positron emission tomography; programmed cell death 1 receptor; radioactive tracers; radionuclide imaging.
Conflict of interest statement
The authors declare no conflict of interest.
Figures


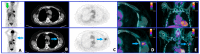

References
-
- Pons-Tostivint E., Latouche A., Vaflard P., Ricci F., Loirat D., Hescot S., Sablin M.-P., Rouzier R., Kamal M., Morel C., et al. Comparative Analysis of Durable Responses on Immune Checkpoint Inhibitors Versus Other Systemic Therapies: A Pooled Analysis of Phase III Trials. JCO Precis. Oncol. 2019;3:1–10. doi: 10.1200/PO.18.00114. - DOI - PubMed
-
- Kanjanapan Y., Day D., Wang L., Al-Sawaihey H., Abbas E., Namini A., Siu L.L., Hansen A., Razak A.A., Spreafico A., et al. Hyperprogressive disease in early-phase immunotherapy trials: Clinical predictors and association with immune-related toxicities. Cancer. 2019;125:1341–1349. doi: 10.1002/cncr.31999. - DOI - PubMed
Publication types
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

