The combination of CTCs and CEA can help guide the management of patients with SPNs suspected of being lung cancer
- PMID: 32041568
- PMCID: PMC7011271
- DOI: 10.1186/s12885-020-6524-1
The combination of CTCs and CEA can help guide the management of patients with SPNs suspected of being lung cancer
Abstract
Objective: Solitary pulmonary nodules (SPNs) is a common radiographic finding and require further evaluation because of the possibility of lung cancer. This study aimed to determine the sensitivity and specificity of circulating tumour cells (CTCs) as a marker for the diagnosis of SPNs and the integration of CTCs, carcinoembryonic antigen (CEA) and imaging findings to improve the sensitivity and specificity of diagnosis in patients with SPNs suspected of being lung cancer.
Method: For the serum biomarker assay, the concentration of CEA was measured by an automated electrochemiluminescence analyzer. CTCs were collected from 6 ml of blood by the SE i-FISH method, which detects the gene copy number in eight chromosomes and the tumour-associated antigen CK18.
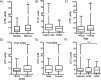
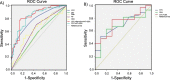
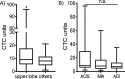
Results: With a threshold of 6 CTC units, the method showed a sensitivity of 67.1% and a specificity of 56.5% in the diagnosis of NSCLC, especially in the upper lobe, in which the diagnostic strength was the highest (P < 0.01). CTCs, CEA and nodule type had the highest diagnostic efficacy (area under the curve, 0.827; 95% confidence interval, 0.752-0.901) in patients with SPNs being suspected lung cancer. Combining CTCs (cut-off value 12 units) with CEA (1.78 ng/ml), the method showed a sensitivity of 77.8% and a specificity of 90% in the diagnosis of NSCLC, especially in the upper lobe, subsolid nodules and nodules ≥8 mm.
Conclusions: Our results demonstrated that CTCs are feasible diagnostic biomarkers in patients with SPNs, especially in the upper lobe. Furthermore, CTCs combined with CEA showed higher diagnostic efficacy in the upper lobe, subsolid nodules and nodules ≥8 mm.
Keywords: CEA; Circulating tumour cells; Lung cancer; Management; Solitary pulmonary nodules.
Conflict of interest statement
The authors declare that they have no competing interests.
Figures
Similar articles
-
Clinical Utility of Circulating Tumor Cells in the Early Detection of Lung Cancer in Patients with a Solitary Pulmonary Nodule.Technol Cancer Res Treat. 2021 Jan-Dec;20:15330338211041465. doi: 10.1177/15330338211041465. Technol Cancer Res Treat. 2021. PMID: 34519585 Free PMC article.
-
Combination of circulating tumor cells with serum carcinoembryonic antigen enhances clinical prediction of non-small cell lung cancer.PLoS One. 2015 May 21;10(5):e0126276. doi: 10.1371/journal.pone.0126276. eCollection 2015. PLoS One. 2015. PMID: 25996878 Free PMC article.
-
[Value of Combined Detection of Cytokines and Tumor Markers in the Differential Diagnosis of Benign and Malignant Solitary Pulmonary Nodules].Zhongguo Fei Ai Za Zhi. 2021 Jun 20;24(6):426-433. doi: 10.3779/j.issn.1009-3419.2021.102.20. Zhongguo Fei Ai Za Zhi. 2021. PMID: 34157802 Free PMC article. Chinese.
-
[Research Progresses of Circulating Tumor Cells in Diagnosis and Treatment of Early Lung Cancer].Zhongguo Fei Ai Za Zhi. 2017 Oct 20;20(10):703-709. doi: 10.3779/j.issn.1009-3419.2017.10.07. Zhongguo Fei Ai Za Zhi. 2017. PMID: 29061218 Free PMC article. Review. Chinese.
-
Diagnostic value of computed tomography scanning in differentiating malignant from benign solitary pulmonary nodules: a meta-analysis.Tumour Biol. 2014 Sep;35(9):8551-8. doi: 10.1007/s13277-014-2113-8. Epub 2014 May 26. Tumour Biol. 2014. PMID: 24859887
Cited by
-
Value of dual-layer spectral detector computed tomography in the diagnosis of benign/malignant solid solitary pulmonary nodules and establishment of a prediction model.Front Oncol. 2023 May 5;13:1147479. doi: 10.3389/fonc.2023.1147479. eCollection 2023. Front Oncol. 2023. PMID: 37213284 Free PMC article.
-
Diagnostic and prognostic role of liquid biopsy in non-small cell lung cancer: evaluation of circulating biomarkers.Explor Target Antitumor Ther. 2020;1(5):343-354. doi: 10.37349/etat.2020.00020. Epub 2020 Oct 30. Explor Target Antitumor Ther. 2020. PMID: 36046486 Free PMC article. Review.
-
Identification of Key Genes in Lung Adenocarcinoma and Establishment of Prognostic Mode.Front Mol Biosci. 2020 Oct 27;7:561456. doi: 10.3389/fmolb.2020.561456. eCollection 2020. Front Mol Biosci. 2020. PMID: 33195408 Free PMC article.
-
Combination of CT and telomerase+ circulating tumor cells improves diagnosis of small pulmonary nodules.JCI Insight. 2021 Jun 8;6(11):e148182. doi: 10.1172/jci.insight.148182. JCI Insight. 2021. PMID: 33905377 Free PMC article.
-
Recent Progress in Enhanced Cancer Diagnosis, Prognosis, and Monitoring Using a Combined Analysis of the Number of Circulating Tumor Cells (CTCs) and Other Clinical Parameters.Cancers (Basel). 2023 Nov 11;15(22):5372. doi: 10.3390/cancers15225372. Cancers (Basel). 2023. PMID: 38001632 Free PMC article. Review.
References
-
- Tamminga M, Groen HH, Hiltermann TJ. Investigating CTCs in NSCLC-a reaction to the study of Jia-Wei wan: a preliminary study on the relationship between circulating tumor cells count and clinical features in patients with non-small cell lung cancer. J Thorac Dis. 2016;8(6):1032–1036. doi: 10.21037/jtd.2016.04.17. - DOI - PMC - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical

