Dynamics of sphingolipids and the serine palmitoyltransferase complex in rat oligodendrocytes during myelination
- PMID: 32041816
- PMCID: PMC7112141
- DOI: 10.1194/jlr.RA120000627
Dynamics of sphingolipids and the serine palmitoyltransferase complex in rat oligodendrocytes during myelination
Abstract
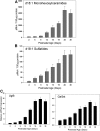
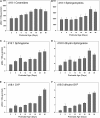
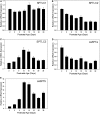
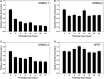
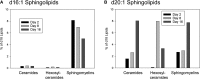
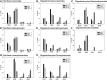
Myelin is a unique lipid-rich membrane structure that accelerates neurotransmission and supports neuronal function. Sphingolipids are critical myelin components. Yet sphingolipid content and synthesis have not been well characterized in oligodendrocytes, the myelin-producing cells of the CNS. Here, using quantitative real-time PCR, LC-MS/MS-based lipid analysis, and biochemical assays, we examined sphingolipid synthesis during the peak period of myelination in the postnatal rat brain. Importantly, we characterized sphingolipid production in isolated oligodendrocytes. We analyzed sphingolipid distribution and levels of critical enzymes and regulators in the sphingolipid biosynthetic pathway, with focus on the serine palmitoyltransferase (SPT) complex, the rate-limiting step in this pathway. During myelination, levels of the major SPT subunits increased and oligodendrocyte maturation was accompanied by extensive alterations in the composition of the SPT complex. These included changes in the relative levels of two alternative catalytic subunits, SPTLC2 and -3, in the relative levels of isoforms of the small subunits, ssSPTa and -b, and in the isoform distribution of the SPT regulators, the ORMDLs. Myelination progression was accompanied by distinct changes in both the nature of the sphingoid backbone and the N-acyl chains incorporated into sphingolipids. We conclude that the distribution of these changes among sphingolipid family members is indicative of a selective channeling of the ceramide backbone toward specific downstream metabolic pathways during myelination. Our findings provide insights into myelin production in oligodendrocytes and suggest how dysregulation of the biosynthesis of this highly specialized membrane could contribute to demyelinating diseases.
Keywords: ORMDL sphingolipid biosynthesis regulator; brain lipids; ceramide; demyelinating disease; developmental myelination; glycolipids; lipidomics; sphingolipids.
Copyright © 2020 Davis et al.
Conflict of interest statement
The authors declare that they have no conflicts of interest with the contents of this article.
Figures




References
-
- Stadelmann C., Timmler S., Barrantes-Freer A., and Simons M.. 2019. Myelin in the central nervous system: structure, function, and pathology. Physiol. Rev. 99: 1381–1431. - PubMed
-
- Norton W. T., and Poduslo S. E.. 1973. Myelination in rat brain: changes in myelin composition during brain maturation. J. Neurochem. 21: 759–773. - PubMed
-
- Bosio A., Binczek E., Haupt W. F., and Stoffel W.. 1998. Composition and biophysical properties of myelin lipid define the neurological defects in galactocerebroside- and sulfatide-deficient mice. J. Neurochem. 70: 308–315. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

