A hemipteran insect reveals new genetic mechanisms and evolutionary insights into tracheal system development
- PMID: 32041884
- PMCID: PMC7049127
- DOI: 10.1073/pnas.1908975117
A hemipteran insect reveals new genetic mechanisms and evolutionary insights into tracheal system development
Abstract
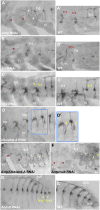
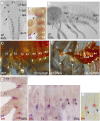
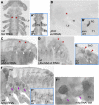
The diversity in the organization of the tracheal system is one of the drivers of insect evolutionary success; however, the genetic mechanisms responsible are yet to be elucidated. Here, we highlight the advantages of utilizing hemimetabolous insects, such as the milkweed bug Oncopeltus fasciatus, in which the final adult tracheal patterning can be directly inferred by examining its blueprint in embryos. By reporting the expression patterns, functions, and Hox gene regulation of trachealess (trh), ventral veinless (vvl), and cut (ct), key genes involved in tracheal development, this study provides important insights. First, Hox genes function as activators, modifiers, and suppressors of trh expression, which in turn results in a difference between the thoracic and abdominal tracheal organization. Second, spiracle morphogenesis requires the input of both trh and ct, where ct is positively regulated by trh As Hox genes regulate trh, we can now mechanistically explain the previous observations of their effects on spiracle formation. Third, the default state of vvl expression in the thorax, in the absence of Hox gene expression, features three lateral cell clusters connected to ducts. Fourth, the exocrine scent glands express vvl and are regulated by Hox genes. These results extend previous findings [Sánchez-Higueras et al., 2014], suggesting that the exocrine glands, similar to the endocrine, develop from the same primordia that give rise to the trachea. The presence of such versatile primordia in the miracrustacean ancestor could account for the similar gene networks found in the glandular and respiratory organs of both insects and crustaceans.
Keywords: Oncopeltus; cut; tracheal system; trachealess; ventral veinless.
Conflict of interest statement
The authors declare no competing interest.
Figures





Similar articles
-
Trachealess (Trh) regulates all tracheal genes during Drosophila embryogenesis.Dev Biol. 2011 Dec 1;360(1):160-72. doi: 10.1016/j.ydbio.2011.09.014. Epub 2011 Sep 22. Dev Biol. 2011. PMID: 21963537 Free PMC article.
-
ventral veinless, a POU domain transcription factor, regulates different transduction pathways required for tracheal branching in Drosophila.Development. 1997 Sep;124(17):3273-81. doi: 10.1242/dev.124.17.3273. Development. 1997. PMID: 9310322
-
An efficient approach to isolate STAT regulated enhancers uncovers STAT92E fundamental role in Drosophila tracheal development.Dev Biol. 2010 Apr 15;340(2):571-82. doi: 10.1016/j.ydbio.2010.02.015. Epub 2010 Feb 18. Dev Biol. 2010. PMID: 20171201 Free PMC article.
-
Hox3/zen and the evolution of extraembryonic epithelia in insects.Adv Exp Med Biol. 2010;689:133-44. doi: 10.1007/978-1-4419-6673-5_10. Adv Exp Med Biol. 2010. PMID: 20795328 Review.
-
Developmental evolution: insights from studies of insect segmentation.Science. 1994 Oct 28;266(5185):581-90. doi: 10.1126/science.7939712. Science. 1994. PMID: 7939712 Review.
Cited by
-
Antioxidant cysteine and methionine derivatives show trachea disruption in insects.PLoS One. 2024 Oct 29;19(10):e0310919. doi: 10.1371/journal.pone.0310919. eCollection 2024. PLoS One. 2024. PMID: 39471169 Free PMC article.
-
Functional analysis of sense organ specification in the Tribolium castaneum larva reveals divergent mechanisms in insects.BMC Biol. 2021 Feb 5;19(1):22. doi: 10.1186/s12915-021-00948-y. BMC Biol. 2021. PMID: 33546687 Free PMC article.
-
Anterior Hox Genes and the Process of Cephalization.Front Cell Dev Biol. 2021 Aug 5;9:718175. doi: 10.3389/fcell.2021.718175. eCollection 2021. Front Cell Dev Biol. 2021. PMID: 34422836 Free PMC article. Review.
-
Specification of the endocrine primordia controlling insect moulting and metamorphosis by the JAK/STAT signalling pathway.PLoS Genet. 2022 Oct 3;18(10):e1010427. doi: 10.1371/journal.pgen.1010427. eCollection 2022 Oct. PLoS Genet. 2022. PMID: 36191039 Free PMC article.
-
Interlocking of co-opted developmental gene networks in Drosophila and the evolution of pre-adaptive novelty.Nat Commun. 2023 Sep 15;14(1):5730. doi: 10.1038/s41467-023-41414-3. Nat Commun. 2023. PMID: 37714829 Free PMC article.
References
-
- Manning G., Krasnow M. A., “Development of the Drosophila tracheal system” in The development of Drosophila Melanogaster, Bate M., Arias A. M., Eds. (Cold Spring Harbor Laboratory Press, Cold Spring Harbor, New York, 1993), pp. 609–685.
-
- Isaac D. D., Andrew D. J., Tubulogenesis in Drosophila: A requirement for the trachealess gene product. Genes Dev. 10, 103–117 (1996). - PubMed
-
- Anderson M. G., Perkins G. L., Chittick P., Shrigley R. J., Johnson W. A., drifter, a Drosophila POU-domain transcription factor, is required for correct differentiation and migration of tracheal cells and midline glia. Genes Dev. 9, 123–137 (1995). - PubMed
-
- de Celis J. F., Llimargas M., Casanova J., Ventral veinless, the gene encoding the Cf1a transcription factor, links positional information and cell differentiation during embryonic and imaginal development in Drosophila melanogaster. Development 121, 3405–3416 (1995). - PubMed
Publication types
MeSH terms
Substances
Associated data
- Actions
- Actions
LinkOut - more resources
Full Text Sources
Research Materials

