Regulation of bone metabolism by megakaryocytes in a paracrine manner
- PMID: 32042021
- PMCID: PMC7010738
- DOI: 10.1038/s41598-020-59250-6
Regulation of bone metabolism by megakaryocytes in a paracrine manner
Abstract
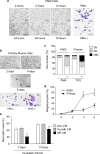
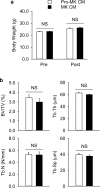
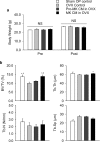
Megakaryocytes (MKs) play key roles in regulating bone metabolism. To test the roles of MK-secreted factors, we investigated whether MK and promegakaryocyte (pro-MK) conditioned media (CM) may affect bone formation and resorption. K562 cell lines were differentiated into mature MKs. Mouse bone marrow macrophages were differentiated into mature osteoclasts, and MC3T3-E1 cells were used for osteoblastic experiments. Bone formation was determined by a calvaria bone formation assay in vivo. Micro-CT analyses were performed in the femurs of ovariectomized female C57B/L6 and Balb/c nude mice after intravenous injections of MK or pro-MK CM. MK CM significantly reduced in vitro bone resorption, largely due to suppressed osteoclastic resorption activity. Compared with pro-MK CM, MK CM suppressed osteoblastic differentiation, but stimulated its proliferation, resulting in stimulation of calvaria bone formation. In ovariectomized mice, treatment with MK CM for 4 weeks significantly increased trabecular bone mass parameters, such as bone volume fraction and trabecular thickness, in nude mice, but not in C57B/L6 mice. In conclusion, MKs may secrete anti-resorptive and anabolic factors that affect bone tissue, providing a novel insight linking MKs and bone cells in a paracrine manner. New therapeutic agents against metabolic bone diseases may be developed from MK-secreted factors.
Conflict of interest statement
The authors declare no competing interests.
Figures



Similar articles
-
S100 Calcium-Binding Protein P Secreted from Megakaryocytes Promotes Osteoclast Maturation.Int J Mol Sci. 2021 Jun 7;22(11):6129. doi: 10.3390/ijms22116129. Int J Mol Sci. 2021. PMID: 34200172 Free PMC article.
-
A novel anti-osteoporotic agent that protects against postmenopausal bone loss by regulating bone formation and bone resorption.Life Sci. 2018 Sep 15;209:409-419. doi: 10.1016/j.lfs.2018.08.014. Epub 2018 Aug 7. Life Sci. 2018. PMID: 30096387
-
The cross-talk between osteoclasts and osteoblasts in response to strontium treatment: involvement of osteoprotegerin.Bone. 2011 Dec;49(6):1290-8. doi: 10.1016/j.bone.2011.08.031. Epub 2011 Sep 9. Bone. 2011. PMID: 21925296
-
A reciprocal regulatory interaction between megakaryocytes, bone cells, and hematopoietic stem cells.Bone. 2006 Nov;39(5):978-984. doi: 10.1016/j.bone.2006.05.019. Epub 2006 Jul 21. Bone. 2006. PMID: 16860008 Review.
-
Megakaryocyte-bone cell interactions.Adv Exp Med Biol. 2010;658:31-41. doi: 10.1007/978-1-4419-1050-9_4. Adv Exp Med Biol. 2010. PMID: 19950013 Review.
Cited by
-
Cellular crosstalk in the bone marrow niche.J Transl Med. 2024 Dec 3;22(1):1096. doi: 10.1186/s12967-024-05900-6. J Transl Med. 2024. PMID: 39627858 Free PMC article. Review.
-
Sphingolipid-Induced Bone Regulation and Its Emerging Role in Dysfunction Due to Disease and Infection.Int J Mol Sci. 2024 Mar 5;25(5):3024. doi: 10.3390/ijms25053024. Int J Mol Sci. 2024. PMID: 38474268 Free PMC article. Review.
-
Megakaryocyte Secreted Factors Regulate Bone Marrow Niche Cells During Skeletal Homeostasis, Aging, and Disease.Calcif Tissue Int. 2023 Jul;113(1):83-95. doi: 10.1007/s00223-023-01095-y. Epub 2023 May 27. Calcif Tissue Int. 2023. PMID: 37243755 Free PMC article. Review.
-
The effects of young and aged, male and female megakaryocyte conditioned media on angiogenic properties of endothelial cells.Aging (Albany NY). 2024 Nov 22;16(21):13181-13200. doi: 10.18632/aging.206077. Epub 2024 Nov 22. Aging (Albany NY). 2024. PMID: 39578050 Free PMC article.
-
S100 Calcium-Binding Protein P Secreted from Megakaryocytes Promotes Osteoclast Maturation.Int J Mol Sci. 2021 Jun 7;22(11):6129. doi: 10.3390/ijms22116129. Int J Mol Sci. 2021. PMID: 34200172 Free PMC article.
References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

