Cancer as a disease of old age: changing mutational and microenvironmental landscapes
- PMID: 32042067
- PMCID: PMC7109142
- DOI: 10.1038/s41416-019-0721-1
Cancer as a disease of old age: changing mutational and microenvironmental landscapes
Abstract
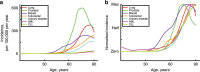
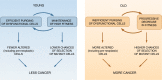
Why do we get cancer mostly when we are old? According to current paradigms, the answer is simple: mutations accumulate in our tissues throughout life, and some of these mutations contribute to cancers. Although mutations are necessary for cancer development, a number of studies shed light on roles for ageing and exposure-dependent changes in tissue landscapes that determine the impact of oncogenic mutations on cellular fitness, placing carcinogenesis into an evolutionary framework. Natural selection has invested in somatic maintenance to maximise reproductive success. Tissue maintenance not only ensures functional robustness but also prevents the occurrence of cancer through periods of likely reproduction by limiting selection for oncogenic events in our cells. Indeed, studies in organisms ranging from flies to humans are revealing conserved mechanisms to eliminate damaged or oncogenically initiated cells from tissues. Reports of the existence of striking numbers of oncogenically initiated clones in normal tissues and of how this clonal architecture changes with age or external exposure to noxious substances provide critical insight into the early stages of cancer development. A major challenge for cancer biology will be the integration of these studies with epidemiology data into an evolutionary theory of carcinogenesis, which could have a large impact on addressing cancer risk and treatment.
Conflict of interest statement
The authors declare no competing interests.
Figures


References
-
- Siegel RL, Miller KD, Jemal A. Cancer statistics, 2018. CA Cancer J. Clin. 2018;68:7–30. - PubMed
-
- Medawar P. An Unsolved Problem of Biology. London: H.K.Lewis; 1952.
Publication types
MeSH terms
Grants and funding
- R01 AG067584/AG/NIA NIH HHS/United States
- 10604/Associazione Italiana per la Ricerca sul Cancro (Italian Association for Cancer Research)/International
- R01 AG067548/AG/NIA NIH HHS/United States
- 18-90-52-DEGR/American Association for Cancer Research (American Association for Cancer Research, Inc.)/International
- I01 BX004495/BX/BLRD VA/United States
LinkOut - more resources
Full Text Sources
Medical

