Chronic irradiation of human cells reduces histone levels and deregulates gene expression
- PMID: 32042076
- PMCID: PMC7010678
- DOI: 10.1038/s41598-020-59163-4
Chronic irradiation of human cells reduces histone levels and deregulates gene expression
Abstract
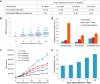
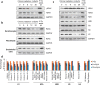
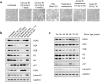
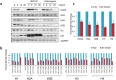
Over the past decades, there have been huge advances in understanding cellular responses to ionising radiation (IR) and DNA damage. These studies, however, were mostly executed with cell lines and mice using single or multiple acute doses of radiation. Hence, relatively little is known about how continuous exposure to low dose ionising radiation affects normal cells and organisms, even though our cells are constantly exposed to low levels of radiation. We addressed this issue by examining the consequences of exposing human primary cells to continuous ionising γ-radiation delivered at 6-20 mGy/h. Although these dose rates are estimated to inflict fewer than a single DNA double-strand break (DSB) per hour per cell, they still caused dose-dependent reductions in cell proliferation and increased cellular senescence. We concomitantly observed histone protein levels to reduce by up to 40%, which in contrast to previous observations, was not mainly due to protein degradation but instead correlated with reduced histone gene expression. Histone reductions were accompanied by enlarged nuclear size paralleled by an increase in global transcription, including that of pro-inflammatory genes. Thus, chronic irradiation, even at low dose-rates, can induce cell senescence and alter gene expression via a hitherto uncharacterised epigenetic route. These features of chronic radiation represent a new aspect of radiation biology.
Conflict of interest statement
The authors declare no competing interests.
Figures

Similar articles
-
In vivo formation of gamma-H2AX and 53BP1 DNA repair foci in blood cells after radioiodine therapy of differentiated thyroid cancer.J Nucl Med. 2010 Aug;51(8):1318-25. doi: 10.2967/jnumed.109.071357. Epub 2010 Jul 21. J Nucl Med. 2010. PMID: 20660387
-
Early and Late Effects of Low-Dose X-ray Exposure in Human Fibroblasts: DNA Repair Foci, Proliferation, Autophagy, and Senescence.Int J Mol Sci. 2024 Jul 28;25(15):8253. doi: 10.3390/ijms25158253. Int J Mol Sci. 2024. PMID: 39125823 Free PMC article.
-
Histone H2AX phosphorylation in normal human cells irradiated with focused ultrasoft X rays: evidence for chromatin movement during repair.Radiat Res. 2006 Jul;166(1 Pt 1):31-8. doi: 10.1667/RR3577.1. Radiat Res. 2006. PMID: 16808616
-
Radiation Induced DNA Double-Strand Breaks in Radiology.Rofo. 2015 Oct;187(10):872-8. doi: 10.1055/s-0035-1553209. Epub 2015 Sep 2. Rofo. 2015. PMID: 26333102 Review.
-
Low-dose radiation-induced responses: focusing on epigenetic regulation.Int J Radiat Biol. 2010 Jul;86(7):517-28. doi: 10.3109/09553001003734592. Int J Radiat Biol. 2010. PMID: 20545569 Review.
Cited by
-
Cell Adhesion Molecules Affected by Ionizing Radiation and Estrogen in an Experimental Breast Cancer Model.Int J Mol Sci. 2022 Oct 21;23(20):12674. doi: 10.3390/ijms232012674. Int J Mol Sci. 2022. PMID: 36293530 Free PMC article.
-
The relationship between epigenetic age and the hallmarks of aging in human cells.Nat Aging. 2022 Jun;2(6):484-493. doi: 10.1038/s43587-022-00220-0. Epub 2022 May 16. Nat Aging. 2022. PMID: 37034474 Free PMC article.
-
Radioprotection and Radiomitigation: From the Bench to Clinical Practice.Biomedicines. 2020 Oct 30;8(11):461. doi: 10.3390/biomedicines8110461. Biomedicines. 2020. PMID: 33142986 Free PMC article. Review.
-
Radiation dose rate effects: what is new and what is needed?Radiat Environ Biophys. 2022 Nov;61(4):507-543. doi: 10.1007/s00411-022-00996-0. Epub 2022 Oct 15. Radiat Environ Biophys. 2022. PMID: 36241855 Free PMC article. Review.
-
Breaking the aging epigenetic barrier.Front Cell Dev Biol. 2022 Jul 28;10:943519. doi: 10.3389/fcell.2022.943519. eCollection 2022. Front Cell Dev Biol. 2022. PMID: 35966762 Free PMC article. Review.
References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous

