Targeting the Mitochondria in Heart Failure: A Translational Perspective
- PMID: 32043022
- PMCID: PMC7000886
- DOI: 10.1016/j.jacbts.2019.07.009
Targeting the Mitochondria in Heart Failure: A Translational Perspective
Abstract
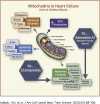
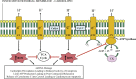
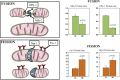
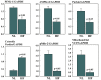
The burden of heart failure (HF) in terms of health care expenditures, hospitalizations, and mortality is substantial and growing. The failing heart has been described as "energy-deprived" and mitochondrial dysfunction is a driving force associated with this energy supply-demand imbalance. Existing HF therapies provide symptomatic and longevity benefit by reducing cardiac workload through heart rate reduction and reduction of preload and afterload but do not address the underlying causes of abnormal myocardial energetic nor directly target mitochondrial abnormalities. Numerous studies in animal models of HF as well as myocardial tissue from explanted failed human hearts have shown that the failing heart manifests abnormalities of mitochondrial structure, dynamics, and function that lead to a marked increase in the formation of damaging reactive oxygen species and a marked reduction in on demand adenosine triphosphate synthesis. Correcting mitochondrial dysfunction therefore offers considerable potential as a new therapeutic approach to improve overall cardiac function, quality of life, and survival for patients with HF.
Keywords: ADP, adenosine diphosphate; ATP, adenosine triphosphate; CI (to V), complex I (to V); Drp, dynamin-related protein; ETC, electron transport chain; HF, heart failure; HFpEF, heart failure with preserved ejection fraction; HFrEF, heart failure with reduced ejection fraction; LV, left ventricular; MPTP, mitochondrial permeability transition pore; Mfn, mitofusin; OPA, optic atrophy; PGC, peroxisome proliferator-activated receptor coactivator; PINK, phosphatase and tensin homolog–inducible kinase; ROS, reactive oxygen species; TAZ, tafazzin; cardiolipin; heart failure; mitochondria; mtDNA, mitochondrial deoxyribonucleic acid; myocardial energetics; oxidative phosphorylation.
© 2020 The Author.
Figures




Similar articles
-
Inefficient Batteries in Heart Failure: Metabolic Bottlenecks Disrupting the Mitochondrial Ecosystem.JACC Basic Transl Sci. 2022 Jul 20;7(11):1161-1179. doi: 10.1016/j.jacbts.2022.03.017. eCollection 2022 Nov. JACC Basic Transl Sci. 2022. PMID: 36687274 Free PMC article. Review.
-
Mitochondrial Morphology, Dynamics, and Function in Human Pressure Overload or Ischemic Heart Disease With Preserved or Reduced Ejection Fraction.Circ Heart Fail. 2019 Feb;12(2):e005131. doi: 10.1161/CIRCHEARTFAILURE.118.005131. Circ Heart Fail. 2019. PMID: 30744415
-
Targeting mitochondrial dysfunction in the treatment of heart failure.Expert Rev Cardiovasc Ther. 2016 Dec;14(12):1305-1313. doi: 10.1080/14779072.2016.1249466. Epub 2016 Oct 31. Expert Rev Cardiovasc Ther. 2016. PMID: 27758146 Review.
-
Abnormalities of Mitochondrial Dynamics in the Failing Heart: Normalization Following Long-Term Therapy with Elamipretide.Cardiovasc Drugs Ther. 2018 Aug;32(4):319-328. doi: 10.1007/s10557-018-6805-y. Cardiovasc Drugs Ther. 2018. PMID: 29951944 Free PMC article.
-
Chronic Therapy With Elamipretide (MTP-131), a Novel Mitochondria-Targeting Peptide, Improves Left Ventricular and Mitochondrial Function in Dogs With Advanced Heart Failure.Circ Heart Fail. 2016 Feb;9(2):e002206. doi: 10.1161/CIRCHEARTFAILURE.115.002206. Circ Heart Fail. 2016. PMID: 26839394 Free PMC article.
Cited by
-
Mitochondrial calcium uniporter complex activation protects against calcium alternans in atrial myocytes.Am J Physiol Heart Circ Physiol. 2020 Oct 1;319(4):H873-H881. doi: 10.1152/ajpheart.00375.2020. Epub 2020 Aug 28. Am J Physiol Heart Circ Physiol. 2020. PMID: 32857593 Free PMC article.
-
Clinical and Molecular Implications of Osteopontin in Heart Failure.Curr Issues Mol Biol. 2022 Aug 11;44(8):3573-3597. doi: 10.3390/cimb44080245. Curr Issues Mol Biol. 2022. PMID: 36005141 Free PMC article. Review.
-
Targeting Mitochondrial Metabolism to Save the Failing Heart.Life (Basel). 2023 Apr 16;13(4):1027. doi: 10.3390/life13041027. Life (Basel). 2023. PMID: 37109556 Free PMC article. Review.
-
Mitochondrial Dysfunction and Heart Disease: Critical Appraisal of an Overlooked Association.Int J Mol Sci. 2021 Jan 9;22(2):614. doi: 10.3390/ijms22020614. Int J Mol Sci. 2021. PMID: 33435429 Free PMC article. Review.
-
Cannabidiol Prevents Heart Failure Dysfunction and Remodeling Through Preservation of Mitochondrial Function and Calcium Handling.JACC Basic Transl Sci. 2025 Jun;10(6):800-821. doi: 10.1016/j.jacbts.2024.12.009. Epub 2025 Feb 19. JACC Basic Transl Sci. 2025. PMID: 40562493 Free PMC article.
References
-
- Barth E., Stammler G., Speiser B., Schaper J. Ultrastructural quantitation of mitochondria and myofilaments in cardiac muscle from 10 different animal species including man. J Mol Cell Cardiol. 1992;24:669–681. - PubMed
-
- Schaper J., Meiser E., Stammler G. Ultrastructural morphometric analysis of myocardium from dogs, rats, hamsters, mice, and from human hearts. Circ Res. 1985;56:377–391. - PubMed
-
- Okonko D.O., Shah A.M. Heart failure: mitochondrial dysfunction and oxidative stress in CHF. Nat Rev Cardiol. 2015;12:6–8. - PubMed
Publication types
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

