Treatment of Pediatric Glioblastoma with Combination Olaparib and Temozolomide Demonstrates 2-Year Durable Response
- PMID: 32043779
- PMCID: PMC7011619
- DOI: 10.1634/theoncologist.2019-0603
Treatment of Pediatric Glioblastoma with Combination Olaparib and Temozolomide Demonstrates 2-Year Durable Response
Abstract
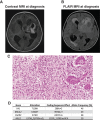
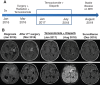
For pediatric patients with high-grade gliomas, standard-of-care treatment includes surgery, chemotherapy, and radiation therapy; however, most patients ultimately succumb to their disease. With advances in genomic characterization of pediatric high-grade gliomas, the use of targeted therapies in combination with current treatment modalities offer the potential to improve survival in this patient population. In this report, we present the case of a 3-year-old girl with glioblastoma who continues to experience an exceptional and durable response (>2 years) to the poly (ADP-ribose) polymerase (PARP) inhibitor olaparib. Our patient presented with persistent and progressive seizure activity that upon workup was the result of a large heterogeneously enhancing, mixed cystic and solid mass in the left frontal-parietal-temporal region. Histopathologic analysis of resected tumor tissue confirmed the diagnosis of glioblastoma, and comprehensive genomic profiling demonstrated absence of any BRAF or H3F3A mutations. Genomic profiling, however, did reveal a probable germline heterozygous BRCA2 Lys3326Ter (K3226*) nonsense variant. After debulking surgery, the patient received standard-of-care treatment with radiation and temozolomide. Nine months later the PARP inhibitor olaparib was administered in combination with temozolomide for 16 cycles. This regimen was well tolerated by the patient and serial imaging showed reduction in tumor size. Since completion of the regimen, the patient remains neurologically intact with no evidence of tumor recurrence. To our knowledge, this represents the first case of a pediatric glioblastoma that maintains a durable response to a therapeutic strategy that included the PARP inhibitor olaparib and more generally highlights the potential clinical utility of incorporating these agents into the treatment of pediatric high-grade gliomas. KEY POINTS: Germline mutations detected in pediatric gliomas may represent a cancer predisposition syndrome. Integrating molecular testing into routine clinical care for pediatric patients with glioma is critical to identify therapeutic targets and patients with a cancer predisposition syndrome. Patients with glioma with defects in DNA repair pathway components (e.g., BRCA1/2) may show increased responsiveness to poly (ADP-ribose) polymerase (PARP) inhibitors. Combining PARP inhibitors with temozolomide (standard-of-care treatment) revealed no adverse events or toxicities over the course of 18 months.
© 2019 The Authors. The Oncologist published by Wiley Periodicals, Inc. on behalf of AlphaMed Press.
Conflict of interest statement
Figures
References
-
- Dodgshun AJ, Sexton‐Oates A, Saffery R et al. Biallelic FANCD1/BRCA2 mutations predisposing to glioblastoma multiforme with multiple oncogenic amplifications. Cancer Genet 2016;209:53–56. - PubMed
-
- Jackson SA, Davis AA, Li J et al. Characteristics of individuals with breast cancer rearrangements in BRCA1 and BRCA2. Cancer 2014;120:1557–1564. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Research Materials
Miscellaneous

