Expression, localization and synthesis of small leucine-rich proteoglycans in developing mouse molar tooth germ
- PMID: 32046476
- PMCID: PMC7029624
- DOI: 10.4081/ejh.2020.3092
Expression, localization and synthesis of small leucine-rich proteoglycans in developing mouse molar tooth germ
Abstract
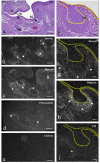
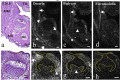
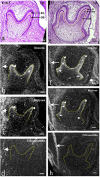
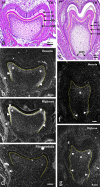
The gene expression and protein synthesis of small leucine-rich proteoglycans (SLRPs), including decorin, biglycan, fibromodulin, and lumican, was analyzed in the context of the hypothesis that they are closely related to tooth formation. In situ hybridization, immunohistochemistry, and organ culture with metabolic labeling of [35S] were carried out in mouse first molar tooth germs of different developmental stages using ICR mice at embryonic day (E) 13.5 to postnatal day (P) 7.0. At the bud and cap stage, decorin mRNA was expressed only in the surrounding mesenchyme, but not within the tooth germ. Biglycan mRNA was then expressed in the condensing mesenchyme and the dental papilla of the tooth germ. At the apposition stage (late bell stage), both decorin and biglycan mRNA were expressed in odontoblasts, resulting in a switch of the pattern of expression within the different stages of odontoblast differentiation. Decorin mRNA was expressed earlier in newly differentiating odontoblasts than biglycan. With odontoblast maturation and dentin formation, decorin mRNA expression was diminished and localized to the newly differentiating odontoblasts at the cervical region. Simultaneously, biglycan mRNA took over and extended its expression throughout the new and mature odontoblasts. Both mRNAs were expressed in the dental pulp underlying the respective odontoblasts. At P7.0, both mRNAs were weakly expressed but maintained their spatial expression patterns. Immunostaining showed that biglycan was localized in the dental papillae and pulp. In addition, all four SLRPs showed clear immunostaining in predentin, although the expressions of fibromodulin and lumican mRNAs were not identified in the tooth germs examined. The organ culture data obtained supported the histological findings that biglycan is more predominant than decorin at the apposition stage. These results were used to identify biglycan as the principal molecule among the SLRPs investigated. Our findings indicate that decorin and biglycan show spatial and temporal differential expressions and play their own tissue-specific roles in tooth development.
Conflict of interest statement
Conflict of interest: The authors declare that they have no competing interests, and all authors confirm accuracy.
Figures


Similar articles
-
Differential expression of decorin and biglycan genes during mouse tooth development.Matrix Biol. 2001 Sep;20(5-6):367-73. doi: 10.1016/s0945-053x(01)00142-1. Matrix Biol. 2001. PMID: 11566271
-
Distribution of biglycan and decorin in rat dental tissue.Braz J Med Biol Res. 2003 Aug;36(8):1061-5. doi: 10.1590/s0100-879x2003000800012. Epub 2003 Jul 23. Braz J Med Biol Res. 2003. PMID: 12886460
-
Comparison of expression patterns between CREB family transcription factor OASIS and proteoglycan core protein genes during murine tooth development.Anat Embryol (Berl). 2003 Apr;206(5):373-80. doi: 10.1007/s00429-003-0311-z. Epub 2003 Mar 21. Anat Embryol (Berl). 2003. PMID: 12684764
-
Proteoglycans and diseases of soft tissues.Adv Exp Med Biol. 2014;802:49-58. doi: 10.1007/978-94-007-7893-1_4. Adv Exp Med Biol. 2014. PMID: 24443020 Review.
-
Small Leucine Rich Proteoglycans (decorin, biglycan and lumican) in cancer.Clin Chim Acta. 2019 Apr;491:1-7. doi: 10.1016/j.cca.2019.01.003. Epub 2019 Jan 7. Clin Chim Acta. 2019. PMID: 30629950 Review.
Cited by
-
mTORC1 signaling pathway regulates tooth repair.Int J Oral Sci. 2023 Mar 16;15(1):14. doi: 10.1038/s41368-023-00218-3. Int J Oral Sci. 2023. PMID: 36927863 Free PMC article.
-
Genome-wide identification of potential odontogenic genes involved in the dental epithelium-mesenchymal interaction during early odontogenesis.BMC Genomics. 2023 Apr 3;24(1):163. doi: 10.1186/s12864-023-09140-8. BMC Genomics. 2023. PMID: 37013486 Free PMC article.
-
The role of vitamin D receptor in predentin mineralization and dental repair after injury.Cell Tissue Res. 2024 Jun;396(3):343-351. doi: 10.1007/s00441-024-03886-7. Epub 2024 Mar 16. Cell Tissue Res. 2024. PMID: 38492000
-
The significant role of glycosaminoglycans in tooth development.Glycobiology. 2024 Apr 19;34(5):cwae024. doi: 10.1093/glycob/cwae024. Glycobiology. 2024. PMID: 38438145 Free PMC article. Review.
-
The vertebrate small leucine-rich proteoglycans: amplification of a clustered gene family and evolution of their transcriptional profile in jawed vertebrates.G3 (Bethesda). 2025 Mar 18;15(3):jkaf003. doi: 10.1093/g3journal/jkaf003. G3 (Bethesda). 2025. PMID: 39774651 Free PMC article.
References
-
- McEwan P, Scott P, Bishop P, Bella J. Structural correlations in the family of small leucine-rich repeat proteins and proteoglycans. J Struct Biol 2006;155:294-305. - PubMed
-
- Orsini G, Ruggeri A, Mazzoni A, Nato F, Manzoli L, Putignano A, et al. A review of the nature, role, and function of dentin non-collagenous proteins. Part 1: proteoglycans and glycoproteins. Endodontic Topics 2012;21:1-18.
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Miscellaneous

