How nasopharyngeal pneumococcal carriage evolved during and after a PCV13-to-PCV10 vaccination programme switch in Belgium, 2016 to 2018
- PMID: 32046817
- PMCID: PMC7014673
- DOI: 10.2807/1560-7917.ES.2020.25.5.1900303
How nasopharyngeal pneumococcal carriage evolved during and after a PCV13-to-PCV10 vaccination programme switch in Belgium, 2016 to 2018
Abstract
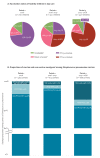
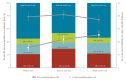
BackgroundThe current carriage study was set up to reinforce surveillance during/after the PCV13-to-PCVC10 switch in Belgium.AimThis observational study monitored carriage of Streptococcus pneumoniae (Sp) serotypes, particularly those no longer covered (3, 6A, 19A), as well as Haemophilus influenzae (Hi), because PCV10 contains the non-typeable Hi protein D.MethodsA total of 2,615 nasopharyngeal swabs from children (6-30 months old) attending day care were collected in three periods over 2016-2018. Children's demographic and clinical characteristics and vaccination status were obtained through a questionnaire. Sp and Hi were identified by culture and PCR. Pneumococcal strains were tested for antimicrobial (non-)susceptibility by disc diffusion and serotyped by Quellung-reaction (Quellung-reaction and PCR for serotypes 3, 6A, 19A).ResultsThe carriage prevalence of Sp (> 75%) remained stable over the successive periods but that of Hi increased (87.4%, 664 Hi-carriers/760 in 2016 vs 93.9%, 895/953 in 2017-2018). The proportion of non-PCV13 vaccine serotypes decreased (94.6%, 438 isolates/463 in 2016 vs 89.7%, 599/668 in 2017-2018) while that of PCV13-non-PCV10 vaccine serotypes (3 + 6A + 19A) increased (0.9%, 4 isolates/463 in 2016 vs 7.8%, 52/668 in 2017-2018), with serotype 19A most frequently identified (87.9%, 58/66 isolates). Non-susceptibility of pneumococci against any of the tested antibiotics was stable over the study period (> 44%).ConclusionsDuring and after the PCV13-to-PCV10 vaccine switch, the proportion of non-PCV13 serotypes decreased, mainly due to a serotype 19A carriage prevalence increase. These results complement invasive pneumococcal disease surveillance data, providing further basis for pneumococcal vaccination programme policy making.
Keywords: Children; Day-care centres; Haemophilus influenzae; Nasopharyngeal carriage; Pneumococcal conjugate vaccines; Streptococcus pneumoniae.
Conflict of interest statement
Figures

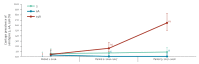
Similar articles
-
Nasopharyngeal s. pneumoniae carriage and density in Belgian infants after 9 years of pneumococcal conjugate vaccine programme.Vaccine. 2018 Jan 2;36(1):15-22. doi: 10.1016/j.vaccine.2017.11.052. Epub 2017 Nov 24. Vaccine. 2018. PMID: 29180027
-
Back to the future? Drastic drop in serotype 19A carriage in daycare centers within two years after a second switch to PCV13 in Belgium.Hum Vaccin Immunother. 2025 Dec;21(1):2484886. doi: 10.1080/21645515.2025.2484886. Epub 2025 Apr 1. Hum Vaccin Immunother. 2025. PMID: 40170143 Free PMC article.
-
Follow-up of serotype distribution and antimicrobial susceptibility of Streptococcus pneumoniae in child carriage after a PCV13-to-PCV10 vaccine switch in Belgium.Vaccine. 2019 Feb 14;37(8):1080-1086. doi: 10.1016/j.vaccine.2018.12.068. Epub 2019 Jan 19. Vaccine. 2019. PMID: 30665775
-
The impact of pneumococcal conjugate vaccines on serotype 19A nasopharyngeal carriage.Expert Rev Vaccines. 2019 Dec;18(12):1243-1270. doi: 10.1080/14760584.2019.1675521. Expert Rev Vaccines. 2019. PMID: 31587592
-
Effects of PCV10 and PCV13 on pneumococcal serotype 6C disease, carriage, and antimicrobial resistance.Vaccine. 2024 Apr 30;42(12):2983-2993. doi: 10.1016/j.vaccine.2024.03.065. Epub 2024 Mar 28. Vaccine. 2024. PMID: 38553292
Cited by
-
Trends in Asymptomatic Nasopharyngeal Streptococcus pneumoniae Carriage with qPCR and Culture Analysis.Microorganisms. 2022 Oct 20;10(10):2074. doi: 10.3390/microorganisms10102074. Microorganisms. 2022. PMID: 36296350 Free PMC article.
-
Clinical and Economic Burden of Pneumococcal Disease Due to Serotypes Contained in Current and Investigational Pneumococcal Conjugate Vaccines in Children Under Five Years of Age.Infect Dis Ther. 2021 Dec;10(4):2701-2720. doi: 10.1007/s40121-021-00544-1. Epub 2021 Oct 11. Infect Dis Ther. 2021. PMID: 34633639 Free PMC article.
-
Detection of SARS-CoV-2 in young children attending day-care centres in Belgium, May 2020 to February 2022.Euro Surveill. 2022 May;27(21):2200380. doi: 10.2807/1560-7917.ES.2022.27.21.2200380. Euro Surveill. 2022. PMID: 35620998 Free PMC article.
-
Clinical and evidence-based considerations for choosing a pneumococcal conjugate vaccine in India: A narrative review.Hum Vaccin Immunother. 2025 Dec;21(1):2482285. doi: 10.1080/21645515.2025.2482285. Epub 2025 Apr 3. Hum Vaccin Immunother. 2025. PMID: 40179380 Free PMC article. Review.
-
Non-invasive Streptococcus pneumoniae infections are associated with different serotypes than invasive infections, Belgium, 2020 to 2023.Euro Surveill. 2024 Nov;29(45):2400108. doi: 10.2807/1560-7917.ES.2024.29.45.2400108. Euro Surveill. 2024. PMID: 39512163 Free PMC article.
References
-
- Satzke C, Dunne EM, Porter BD, Klugman KP, Mulholland EK, PneuCarriage project group The PneuCarriage Project: A Multi-Centre Comparative Study to Identify the Best Serotyping Methods for Examining Pneumococcal Carriage in Vaccine Evaluation Studies. PLoS Med. 2015;12(11):e1001903 , discussion e1001903. 10.1371/journal.pmed.1001903 - DOI - PMC - PubMed
-
- World Health Organization (WHO) Pneumococcal conjugate vaccines in infants and children under 5 years of age: WHO position paper - February 2019. WER. 2019;94(8):85-104.
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
