Alzheimer's disease: targeting the glutamatergic system
- PMID: 32048098
- PMCID: PMC7196085
- DOI: 10.1007/s10522-020-09860-4
Alzheimer's disease: targeting the glutamatergic system
Abstract
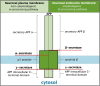
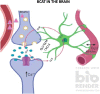
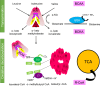
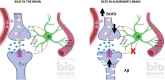
Alzheimer's disease (AD) is a debilitating neurodegenerative disease that causes a progressive decline in memory, language and problem solving. For decades mechanism-based therapies have primarily focused on amyloid β (Aβ) processing and pathways that govern neurofibrillary tangle generation. With the potential exception to Aducanumab, a monotherapy to target Aβ, clinical trials in these areas have been challenging and have failed to demonstrate efficacy. Currently, the prescribed therapies for AD are those that target the cholinesterase and glutamatergic systems that can moderately reduce cognitive decline, dependent on the individual. In the brain, over 40% of neuronal synapses are glutamatergic, where the glutamate level is tightly regulated through metabolite exchange in neuronal, astrocytic and endothelial cells. In AD brain, Aβ can interrupt effective glutamate uptake by astrocytes, which evokes a cascade of events that leads to neuronal swelling, destruction of membrane integrity and ultimately cell death. Much work has focussed on the post-synaptic response with little insight into how glutamate is regulated more broadly in the brain and the influence of anaplerotic pathways that finely tune these mechanisms. The role of blood branched chain amino acids (BCAA) in regulating neurotransmitter profiles under disease conditions also warrant discussion. Here, we review the importance of the branched chain aminotransferase proteins in regulating brain glutamate and the potential consequence of dysregulated metabolism in the context of BCAA or glutamate accumulation. We explore how the reported benefits of BCAA supplementation or restriction in improving cognitive function in other neurological diseases may have potential application in AD. Given that memantine, the glutamate receptor agonist, shows clinical relevance it is now timely to research related pathways, an understanding of which could identify novel approaches to treatment of AD.
Keywords: Aging; Alzheimer's disease; BCAT; Branched chain amino acids; Glutamate.
Figures
Similar articles
-
Alzheimer's disease.Subcell Biochem. 2012;65:329-52. doi: 10.1007/978-94-007-5416-4_14. Subcell Biochem. 2012. PMID: 23225010 Review.
-
Effects of ketone bodies in Alzheimer's disease in relation to neural hypometabolism, β-amyloid toxicity, and astrocyte function.J Neurochem. 2015 Jul;134(1):7-20. doi: 10.1111/jnc.13107. Epub 2015 Apr 23. J Neurochem. 2015. PMID: 25832906 Review.
-
Astrocytic adenosine A2A receptors control the amyloid-β peptide-induced decrease of glutamate uptake.J Alzheimers Dis. 2012;31(3):555-67. doi: 10.3233/JAD-2012-120469. J Alzheimers Dis. 2012. PMID: 22647260
-
Reactive Astrocytes as Drug Target in Alzheimer's Disease.Biomed Res Int. 2018 May 14;2018:4160247. doi: 10.1155/2018/4160247. eCollection 2018. Biomed Res Int. 2018. PMID: 29888263 Free PMC article. Review.
-
Recent Insights on Glutamatergic Dysfunction in Alzheimer's Disease and Therapeutic Implications.Neuroscientist. 2023 Aug;29(4):461-471. doi: 10.1177/10738584211069897. Epub 2022 Jan 25. Neuroscientist. 2023. PMID: 35073787 Review.
Cited by
-
Metabolomic Signatures of Alzheimer's Disease Indicate Brain Region-Specific Neurodegenerative Progression.Int J Mol Sci. 2023 Sep 30;24(19):14769. doi: 10.3390/ijms241914769. Int J Mol Sci. 2023. PMID: 37834217 Free PMC article.
-
Disease-Associated Neurotoxic Astrocyte Markers in Alzheimer Disease Based on Integrative Single-Nucleus RNA Sequencing.Cell Mol Neurobiol. 2024 Feb 12;44(1):20. doi: 10.1007/s10571-024-01453-w. Cell Mol Neurobiol. 2024. PMID: 38345650 Free PMC article.
-
The dopaminergic system and Alzheimer's disease.Neural Regen Res. 2025 Sep 1;20(9):2495-2512. doi: 10.4103/NRR.NRR-D-24-00230. Epub 2024 Sep 24. Neural Regen Res. 2025. PMID: 39314145 Free PMC article.
-
Natural Products and Their Neuroprotective Effects in Degenerative Brain Diseases: A Comprehensive Review.Int J Mol Sci. 2024 Oct 18;25(20):11223. doi: 10.3390/ijms252011223. Int J Mol Sci. 2024. PMID: 39457003 Free PMC article. Review.
-
Spatio-temporal metabolic rewiring in the brain of TgF344-AD rat model of Alzheimer's disease.Sci Rep. 2022 Oct 10;12(1):16958. doi: 10.1038/s41598-022-20962-6. Sci Rep. 2022. PMID: 36216838 Free PMC article.
References
-
- Albin RL, Greenamyre JT. Alternative excitotoxic hypotheses. Neurology. 1992;42(4):733–738. - PubMed
-
- Antuono PG, Jones JL, Wang Y, Li SJ. Decreased glutamate+glutamine in Alzheimer's disease detected in vivo with (1)H-MRS at 0.5 T. Neurology. 2001;56(6):737–742. - PubMed
-
- Aquilani R, Boselli M, Boschi F, Viglio S, Iadarola P, Dossena M, Pastoris O, Verri M. Branched-chain amino acids may improve recovery from a vegetative or minimally conscious state in patients with traumatic brain injury: a pilot study. Arch Phys Med Rehabil. 2008;89(9):1642–1647. - PubMed
-
- Aquilani R, Scocchi M, Iadarola P, Franciscone P, Verri M, Boschi F, Pasini E, Viglio S. Protein supplementation may enhance the spontaneous recovery of neurological alterations in patients with ischaemic stroke. Clin Rehabil. 2008;22(12):1042–1050. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

