Rapid Donor Identification Improves Survival in High-Risk First-Remission Patients With Acute Myeloid Leukemia
- PMID: 32048933
- PMCID: PMC7291544
- DOI: 10.1200/JOP.19.00133
Rapid Donor Identification Improves Survival in High-Risk First-Remission Patients With Acute Myeloid Leukemia
Erratum in
-
Erratum: Rapid Donor Identification Improves Survival in High-Risk First-Remission Patients With Acute Myeloid Leukemia.JCO Oncol Pract. 2025 Mar 20:OP2500135. doi: 10.1200/OP-25-00135. Online ahead of print. JCO Oncol Pract. 2025. PMID: 40112240 No abstract available.
Abstract
Purpose: Patients with acute myeloid leukemia with high-risk cytogenetics in first complete remission (CR1) achieve better outcomes if they undergo allogeneic hematopoietic cell transplantation (HCT) compared with consolidation chemotherapy alone. However, only approximately 40% of such patients typically proceed to HCT.
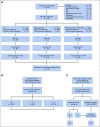
Methods: We used a prospective organized approach to rapidly identify donors to improve the allogeneic HCT rate in adults with high-risk acute myeloid leukemia in CR1. Newly diagnosed patients had cytogenetics obtained at enrollment, and those with high-risk cytogenetics underwent expedited HLA typing and were encouraged to be referred for consultation with a transplantation team with the goal of conducting an allogeneic HCT in CR1.
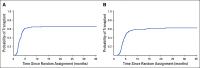
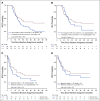
Results: Of 738 eligible patients (median age, 49 years; range, 18-60 years of age), 159 (22%) had high-risk cytogenetics and 107 of these patients (67%) achieved CR1. Seventy (65%) of the high-risk patients underwent transplantation in CR1 (P < .001 compared with the historical rate of 40%). Median time to HCT from CR1 was 77 days (range, 20-356 days). In landmark analysis, overall survival (OS) among patients who underwent transplantation was significantly better compared with that of patients who did not undergo transplantation (2-year OS, 48% v 35%, respectively [P = .031]). Median relapse-free survival after transplantation in the high-risk cohort who underwent transplantation in CR1 (n = 70) was 11.5 months (range, 4-47 months), and median OS after transplantation was 14 months (range, 4-44 months).
Conclusion: Early cytogenetic testing with an organized effort to identify a suitable allogeneic HCT donor led to a CR1 transplantation rate of 65% in the high-risk group, which, in turn, led to an improvement in OS when compared with the OS of patients who did not undergo transplantation.
Figures
References
-
- Slovak ML, Kopecky KJ, Cassileth PA, et al. Karyotypic analysis predicts outcome of preremission and postremission therapy in adult acute myeloid leukemia: A Southwest Oncology Group/Eastern Cooperative Oncology Group Study. Blood. 2000;96:4075–4083. - PubMed
-
- Byrd JC, Mrózek K, Dodge RK, et al. Pretreatment cytogenetic abnormalities are predictive of induction success, cumulative incidence of relapse, and overall survival in adult patients with de novo acute myeloid leukemia: Results from Cancer and Leukemia Group B (CALGB 8461) Blood. 2002;100:4325–4336. - PubMed
-
- Peniket A, Wainscoat J, Side L, et al. Del (9q) AML: Clinical and cytological characteristics and prognostic implications. Br J Haematol. 2005;129:210–220. - PubMed
-
- Middeke JM, Fang M, Cornelissen JJ, et al. Outcome of patients with abnl(17p) acute myeloid leukemia after allogeneic hematopoietic stem cell transplantation. Blood. 2014;123:2960–2967. - PubMed
Publication types
MeSH terms
Grants and funding
- UG1 CA189856/CA/NCI NIH HHS/United States
- U10 CA046113/CA/NCI NIH HHS/United States
- U10 CA180798/CA/NCI NIH HHS/United States
- UG1 CA189830/CA/NCI NIH HHS/United States
- U10 CA073590/CA/NCI NIH HHS/United States
- UG1 CA189853/CA/NCI NIH HHS/United States
- U10 CA180855/CA/NCI NIH HHS/United States
- U10 CA180820/CA/NCI NIH HHS/United States
- U10 CA180888/CA/NCI NIH HHS/United States
- U10 CA180816/CA/NCI NIH HHS/United States
- UG1 CA233339/CA/NCI NIH HHS/United States
- N01 CA004919/CA/NCI NIH HHS/United States
- U10 CA180826/CA/NCI NIH HHS/United States
- UG1 CA189971/CA/NCI NIH HHS/United States
- U10 CA012644/CA/NCI NIH HHS/United States
- U10 CA016385/CA/NCI NIH HHS/United States
- U10 CA004919/CA/NCI NIH HHS/United States
- N01 CA013612/CA/NCI NIH HHS/United States
- U10 CA077202/CA/NCI NIH HHS/United States
- U10 CA013612/CA/NCI NIH HHS/United States
- U10 CA180835/CA/NCI NIH HHS/United States
- UG1 CA233330/CA/NCI NIH HHS/United States
- U10 CA180821/CA/NCI NIH HHS/United States
- U10 CA180801/CA/NCI NIH HHS/United States
- U10 CA180858/CA/NCI NIH HHS/United States
- UG1 CA189848/CA/NCI NIH HHS/United States
- UG1 CA189872/CA/NCI NIH HHS/United States
- UG1 CA189822/CA/NCI NIH HHS/United States
- N01 CA035119/CA/NCI NIH HHS/United States
- U10 CA180846/CA/NCI NIH HHS/United States
- UG1 CA189858/CA/NCI NIH HHS/United States
- UG1 CA189860/CA/NCI NIH HHS/United States
- U10 CA046282/CA/NCI NIH HHS/United States
- U10 CA180863/CA/NCI NIH HHS/United States
- U10 CA035119/CA/NCI NIH HHS/United States
- U10 CA011083/CA/NCI NIH HHS/United States
- UG1 CA189957/CA/NCI NIH HHS/United States
- U10 CA046368/CA/NCI NIH HHS/United States
- U10 CA180819/CA/NCI NIH HHS/United States
- UG1 CA189953/CA/NCI NIH HHS/United States
- UG1 CA233328/CA/NCI NIH HHS/United States
LinkOut - more resources
Full Text Sources
Medical
Research Materials

