BDNF as a Promising Therapeutic Agent in Parkinson's Disease
- PMID: 32050617
- PMCID: PMC7037114
- DOI: 10.3390/ijms21031170
BDNF as a Promising Therapeutic Agent in Parkinson's Disease
Abstract
Brain-derived neurotrophic factor (BDNF) promotes neuroprotection and neuroregeneration. In animal models of Parkinson's disease (PD), BDNF enhances the survival of dopaminergic neurons, improves dopaminergic neurotransmission and motor performance. Pharmacological therapies of PD are symptom-targeting, and their effectiveness decreases with the progression of the disease; therefore, new therapeutical approaches are needed. Since, in both PD patients and animal PD models, decreased level of BDNF was found in the nigrostriatal pathway, it has been hypothesized that BDNF may serve as a therapeutic agent. Direct delivery of exogenous BDNF into the patient's brain did not relieve the symptoms of disease, nor did attempts to enhance BDNF expression with gene therapy. Physical training was neuroprotective in animal models of PD. This effect is mediated, at least partly, by BDNF. Animal studies revealed that physical activity increases BDNF and tropomyosin receptor kinase B (TrkB) expression, leading to inhibition of neurodegeneration through induction of transcription factors and expression of genes related to neuronal proliferation, survival, and inflammatory response. This review focuses on the evidence that increasing BDNF level due to gene modulation or physical exercise has a neuroprotective effect and could be considered as adjunctive therapy in PD.
Keywords: PD therapy; Parkinson’s disease; brain-derived neurotrophic factor; neurodegeneration; neuroprotection; physical exercise.
Conflict of interest statement
The authors declare no conflict of interest.
Figures

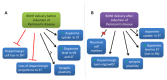

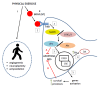
References
-
- Kerschensteiner M., Gallmeier E., Behrens L., Leal V.V., Misgeld T., Klinkert W.E.F., Kolbeck R., Hoppe E., Oropeza-Wekerle R.-L., Bartke I., et al. Activated Human T Cells, B Cells, and Monocytes Produce Brain-derived Neurotrophic Factor In Vitro and in Inflammatory Brain Lesions: A Neuroprotective Role of Inflammation? J. Exp. Med. 1999;189:865–870. doi: 10.1084/jem.189.5.865. - DOI - PMC - PubMed
-
- Donovan M.J., Miranda R.C., Kraemer R., McCaffrey T.A., Tessarollo L., Mahadeo D., Sharif S., Kaplan D.R., Tsoulfas P., Parada L., et al. Neurotrophin and neurotrophin receptors in vascular smooth muscle cells: Regulation of expression in response to injury. Am. J. Pathol. 1995;147:309–324. - PMC - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

