Caspase-1 interdomain linker cleavage is required for pyroptosis
- PMID: 32051255
- PMCID: PMC7025033
- DOI: 10.26508/lsa.202000664
Caspase-1 interdomain linker cleavage is required for pyroptosis
Abstract
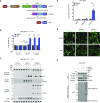
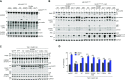
Pathogen-related signals induce a number of cytosolic pattern-recognition receptors (PRRs) to form canonical inflammasomes, which activate pro-caspase-1 and trigger pyroptotic cell death. All well-studied inflammasome-forming PRRs oligomerize with the adapter protein ASC (apoptosis-associated speck-like protein containing a CARD) to generate a large structure in the cytosol, which induces the dimerization, autoproteolysis, and activation of the pro-caspase-1 zymogen. However, several PRRs can also directly interact with pro-caspase-1 without ASC, forming smaller "ASC-independent" inflammasomes. It is currently thought that little, if any, pro-caspase-1 autoproteolysis occurs during, and is not required for, ASC-independent inflammasome signaling. Here, we show that the related human PRRs NLRP1 and CARD8 exclusively form ASC-dependent and ASC-independent inflammasomes, respectively, identifying CARD8 as the first canonical inflammasome-forming PRR that does not form an ASC-containing signaling platform. Despite their different structures, we discovered that both the NLRP1 and CARD8 inflammasomes require pro-caspase-1 autoproteolysis between the small and large catalytic subunits to induce pyroptosis. Thus, pro-caspase-1 self-cleavage is a required regulatory step for pyroptosis induced by human canonical inflammasomes.
© 2020 Ball et al.
Conflict of interest statement
The authors declare that they have no conflict of interest.
Figures






References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Miscellaneous
