Tertiary amine-directed and involved carbonylative cyclizations through Pd/Cu-cocatalyzed multiple C-X (X = H or N) bond cleavage
- PMID: 32055315
- PMCID: PMC7003976
- DOI: 10.1039/c9sc03081f
Tertiary amine-directed and involved carbonylative cyclizations through Pd/Cu-cocatalyzed multiple C-X (X = H or N) bond cleavage
Abstract
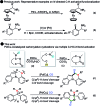
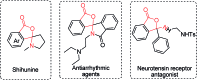
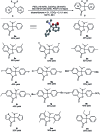
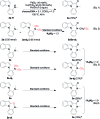
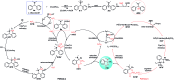
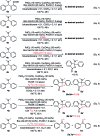
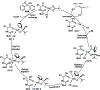
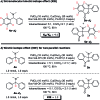
A novel Pd/Cu-cocatalyzed carbonylative cyclization by C-H activation and N-dealkylative C-N bond activation has been developed for the chemoselective construction of synthetically useful heterocycles. The N,N-dimethylamine group on o-indolyl-N,N-dimethylarylamines was found to act as both the directing group and reactive component in this C-H carbonylative cyclization reaction. Furthermore, a unique C-H oxidation/carbonylative lactonization of diarylmethylamines is firstly demonstrated under modified reaction conditions, which could be easily applicable to the one-step synthesis of multi-substituted phthalides bearing an N,O-ketal skeleton that is difficult to access by previously reported methods. Mechanistic studies implicate that Pd/Cu-cocatalyzed C-H oxidation/carbonylative lactonization is a sequential reaction system via Cu-catalyzed C(sp3)-H oxidation and Pd-catalyzed oxidative carbonylation of the C(sp2)-H bond. It was found that trace amounts of water are essential to promote the Cu-catalyzed C(sp3)-H oxidation of diarylmethylamine for the formation of the hydroxyl group, which could act as an in situ-formed directing group in the intramolecular carbonylative lactonization step.
This journal is © The Royal Society of Chemistry 2019.
Figures

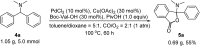
Similar articles
-
Natural product syntheses via carbonylative cyclizations.Nat Prod Rep. 2019 Jan 1;36(1):174-219. doi: 10.1039/c8np00033f. Epub 2018 Jun 20. Nat Prod Rep. 2019. PMID: 29923586 Review.
-
Copper-Catalyzed Carbonylative Coupling of Alkyl Halides.Acc Chem Res. 2021 May 4;54(9):2261-2274. doi: 10.1021/acs.accounts.1c00115. Epub 2021 Apr 21. Acc Chem Res. 2021. PMID: 33881839
-
Pd-Catalyzed Aromatic Dual C-H Acylations and Intramolecular Cyclization: Access to Quinoline-Substituted Hydroxyl Isoindolones.Molecules. 2024 Nov 15;29(22):5397. doi: 10.3390/molecules29225397. Molecules. 2024. PMID: 39598786 Free PMC article.
-
Copper-Catalyzed Carbonylative Cyclization of CO2: A Promising Approach for Synthesis of Flavone.Adv Sci (Weinh). 2025 Apr;12(13):e2415795. doi: 10.1002/advs.202415795. Epub 2025 Feb 7. Adv Sci (Weinh). 2025. PMID: 39921264 Free PMC article.
-
Metal-catalyzed silylation of sp3C-H bonds.Chem Soc Rev. 2021 Apr 26;50(8):5062-5085. doi: 10.1039/d0cs01392g. Chem Soc Rev. 2021. PMID: 33629997 Review.
Cited by
-
Carbonylative synthesis and functionalization of indoles.Beilstein J Org Chem. 2024 Apr 30;20:973-1000. doi: 10.3762/bjoc.20.87. eCollection 2024. Beilstein J Org Chem. 2024. PMID: 38711593 Free PMC article. Review.
References
-
-
For recent examples on the C–H functionalization, see:
- Berger F., Plutschack M. B., Riegger J., Yu M. W., Speicher S., Ho M., Frank N., Ritter T. Nature. 2019;567:223. - PubMed
- Zhang X., Lu G., Sun M., Mahankali M., Ma Y., Zhang M., Hua W., Hu Y., Wang Q., Chen J., He G., Qi X., Sheng W., Liu P., Chen G. Nat. Chem. 2018;10:540. - PubMed
- Kinney R. G., Tjutrins J., Torres G. M., Liu N. J., Kulkarni O., Arndtsen B. A. Nat. Chem. 2018;10:193. - PubMed
- Wang P., Verma P., Xia G., Shi J., Qiao J. X., Tao S., Cheng P. T. W., Poss M. A., Farmer M. E., Yeung K.-S., Yu J.-Q. Nature. 2017;551:489. - PMC - PubMed
- Liao K., Negretti S., Musaev D. G., Bacsa J., Davies H. M. Nature. 2016;533:230. - PubMed
- Chu J. C. K., Rovis T. Nature. 2016;539:272. - PMC - PubMed
- Willcox D., Chappell B. G. N., Hogg K. F., Calleja J., Smalley A. P., Gaunt M. J. Science. 2016;354:851. - PubMed
- Zhang F. L., Hong K., Li T. J., Park H., Yu J.-Q. Science. 2016;351:252. - PMC - PubMed
- Shaw M. H., Shurtleff V. M., Terrett J. A., Cuthbertson J. D., Macmillan D. W. C. Science. 2016;352:1304. - PMC - PubMed
- Cook A. K., Schimler S. D., Matzger A. J., Sanford M. S. Science. 2016;351:1421. - PubMed
- Shao B., Bagdasarian A. L., Popov S., Nelson H. M. Science. 2017;355:1403. - PubMed
- Li X., Li X., Jiao N. J. Am. Chem. Soc. 2015;137:9246. - PubMed
- Xiao B., Gong T.-J., Liu Z.-J., Liu J.-H., Luo D.-F., Xu J., Liu L. J. Am. Chem. Soc. 2011;133:9250. - PubMed
-
-
-
For selected reviews on C–H functionalization, see:
- Williamson J. B., Lewis S. E., Johnson III R. R., Manning I. M., Leibfarth F. A. Angew. Chem., Int. Ed. 2019;58:8654. - PubMed
- Abrams D. J., Provencher P. A., Sorensen E. J. Chem. Soc. Rev. 2018;47:8925. - PubMed
- Davies H. M. L., Morton D. ACS Cent. Sci. 2017;3:936. - PMC - PubMed
- Murakami K., Yamada S., Kaneda T., Itami K. Chem. Rev. 2017;117:9302. - PubMed
- Davies H. M. L., Morton D. J. Org. Chem. 2016;81:343. - PubMed
- Doyle M. P., Goldberg K. I. Acc. Chem. Res. 2012;45:777. - PubMed
- Yamaguchi J., Yamaguchi A. D., Itami K. Angew. Chem., Int. Ed. 2012;51:8960. - PubMed
- Davies H. M. L., Bois J. D., Yu J.-Q. Chem. Soc. Rev. 2011;40:1855. - PubMed
- Gutekunst W. R., Baran P. S. Chem. Soc. Rev. 2011;40:1976. - PubMed
- Wu X.-F., Neumann H., Beller M. ChemSusChem. 2013;6:229. - PubMed
- Gabriele B., Mancuso R., Salerno G. Eur. J. Org. Chem. 2012:6825.
-
-
-
For selected reviews, see:
- Gandeepan P., Ackermann L. Chem. 2018;4:199.
- Ma C., Fang P., Mei T.-S. ACS Catal. 2018;8:7179.
- Ye W., Hu P., Zhang M., Su W. Chem. Rev. 2017;117:8864. - PubMed
- He J., Wasa M., Chan K. S. L., Shao Q., Yu J.-Q. Chem. Rev. 2017;117:8754. - PMC - PubMed
- Labinger J. A. Chem. Rev. 2017;117:8483. - PubMed
- Guo X.-X., Gu D.-W., Wu Z., Zhang W. Chem. Rev. 2015;115:1622. - PubMed
- Ye B., Cramer N. Acc. Chem. Res. 2015;48:1308. - PubMed
- Girard S. A., Knauber T., Li C.-J. Angew. Chem., Int. Ed. 2014;53:74. - PubMed
- Rouquet G., Chatani N. Angew. Chem., Int. Ed. 2013;52:11726. - PubMed
- Neufeldt S. R., Sanford M. S. Acc. Chem. Res. 2012;45:936. - PMC - PubMed
- Engle K. M., Mei T.-S., Wasa M., Yu J.-Q. Acc. Chem. Res. 2012;45:788. - PMC - PubMed
- Kuhl N., Hopkinson M. N., Wencel-Delord J., Glorius F. Angew. Chem., Int. Ed. 2012;51:10236. - PubMed
- Arockiam P. B., Bruneau C., Dixneuf P. H. Chem. Rev. 2012;112:5879. - PubMed
- Liu C., Zhang H., Shi W., Lei A. Chem. Rev. 2011;111:1780. - PubMed
- Hartwig J. F. Chem. Soc. Rev. 2011;40:1992. - PubMed
- Yeung C. S., Dong V. M. Chem. Rev. 2011;111:1215. - PubMed
- Lyons T. W., Sanford M. S. Chem. Rev. 2010;110:1147. - PMC - PubMed
- Chen X., Engle K. M., Wang D.-H., Yu J.-Q. Angew. Chem., Int. Ed. 2009;48:5094. - PMC - PubMed
- Daugulis O., Do H.-Q., Shabashov D. Acc. Chem. Res. 2009;42:1074. - PMC - PubMed
- Ackermann L., Vicente R., Kapdi A. R. Angew. Chem., Int. Ed. 2009;48:9792. - PubMed
- Giri R., Shi B.-F., Engle K. M., Maugel N., Yu J.-Q. Chem. Soc. Rev. 2009;38:3242. - PubMed
- Davies H. M. L., Manning J. R. Nature. 2008;451:417. - PMC - PubMed
- Sambiagio C., Schonbauer D., Blieck R., Dao-Huy T., Pototschnig G., Schaaf P., Wiesinger T., Zia M. F., Wencel-Delord J., Besset T., Maes B. U. W., Schnurch M. Chem. Soc. Rev. 2018;47:6603. - PMC - PubMed
-
-
- Zaitsev V. G., Shabashov D., Daugulis O. J. Am. Chem. Soc. 2005;127:13154. - PubMed
- Reddy B. V. S., Reddy L. R., Corey E. J. Org. Lett. 2006;8:3391. - PubMed
- Cheng X.-F., Li Y., Su Y. M., Yin F., Wang J.-Y., Sheng J., Vora H. U., Wang X. S., Yu J.-Q. J. Am. Chem. Soc. 2013;135:1236. - PMC - PubMed
- Gallardo-Donaire J., Martin R. J. Am. Chem. Soc. 2013;135:9350. - PubMed
- Luo S., Luo F.-X., Zhang X.-S., Shi Z.-J. Angew. Chem., Int. Ed. 2013;52:10598. - PubMed
- Lee T.-H., Jayakumar J., Cheng C.-H., Chuang S.-C. Chem. Commun. 2013;49:11797. - PubMed
-
-
For selected reports, see:
- Cai Z.-J., Liu C.-X., Gu Q., Zheng C., You S.-L. Angew. Chem., Int. Ed. 2019;58:2149. - PubMed
- Plevova K., Mudrakova B., Rakovsky E., Sebesta R. J. Org. Chem. 2019;84:7312. - PubMed
- Zou X., Zhao H., Li Y., Gao Q., Ke Z., Xu S. J. Am. Chem. Soc. 2019;141:5334. - PubMed
- Cai G., Fu Y., Li Y., Wan X., Shi Z.-J. J. Am. Chem. Soc. 2007;129:7666. - PubMed
- Li H., Cai G.-X., Shi Z.-J. Dalton Trans. 2010;39:10442. - PubMed
- Liu B., Shi B. F. Synlett. 2013;24:2274.
- Wang Z., Li Y., Zhu F., Wu X.-F. Adv. Synth. Catal. 2016;358:2855.
-
LinkOut - more resources
Full Text Sources

