The antiarrhythmic compound efsevin directly modulates voltage-dependent anion channel 2 by binding to its inner wall and enhancing mitochondrial Ca2+ uptake
- PMID: 32059260
- PMCID: PMC7279994
- DOI: 10.1111/bph.15022
The antiarrhythmic compound efsevin directly modulates voltage-dependent anion channel 2 by binding to its inner wall and enhancing mitochondrial Ca2+ uptake
Abstract
Background and purpose: The synthetic compound efsevin was recently identified to suppress arrhythmogenesis in models of cardiac arrhythmia, making it a promising candidate for antiarrhythmic therapy. Its activity was shown to be dependent on the voltage-dependent anion channel 2 (VDAC2) in the outer mitochondrial membrane. Here, we investigated the molecular mechanism of the efsevin-VDAC2 interaction.
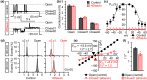
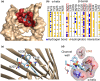
Experimental approach: To evaluate the functional interaction of efsevin and VDAC2, we measured currents through recombinant VDAC2 in planar lipid bilayers. Using molecular ligand-protein docking and mutational analysis, we identified the efsevin binding site on VDAC2. Finally, physiological consequences of the efsevin-induced modulation of VDAC2 were analysed in HL-1 cardiomyocytes.
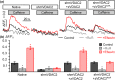
Key results: In lipid bilayers, efsevin reduced VDAC2 conductance and shifted the channel's open probability towards less anion-selective closed states. Efsevin binds to a binding pocket formed by the inner channel wall and the pore-lining N-terminal α-helix. Exchange of amino acids N207, K236 and N238 within this pocket for alanines abolished the channel's efsevin-responsiveness. Upon heterologous expression in HL-1 cardiomyocytes, both channels, wild-type VDAC2 and the efsevin-insensitive VDAC2AAA restored mitochondrial Ca2+ uptake, but only wild-type VDAC2 was sensitive to efsevin.
Conclusion and implications: In summary, our data indicate a direct interaction of efsevin with VDAC2 inside the channel pore that leads to modified gating and results in enhanced SR-mitochondria Ca2+ transfer. This study sheds new light on the function of VDAC2 and provides a basis for structure-aided chemical optimization of efsevin.
© 2020 The Authors. British Journal of Pharmacology published by John Wiley & Sons Ltd on behalf of British Pharmacological Society.
Conflict of interest statement
The authors declare no conflicts of interest.
Figures


References
-
- Benjamin, E. J. , Virani, S. S. , Callaway, C. W. , Chamberlain, A. M. , Chang, A. R. , Cheng, S. , … American Heart Association Council on Epidemiology and Prevention Statistics Committee and Stroke Statistics Subcommittee . (2018). Heart disease and stroke statistics—2018 update: A report from the American Heart Association. Circulation, 137, e67–e492. 10.1161/CIR.0000000000000558 - DOI - PubMed
-
- Claycomb, W. C. , Lanson, N. A. , Stallworth, B. S. , Egeland, D. B. , Delcarpio, J. B. , Bahinski, A. , & Izzo, N. J. (1998). HL‐1 cells: A cardiac muscle cell line that contracts and retains phenotypic characteristics of the adult cardiomyocyte. Proceedings of the National Academy of Sciences of the United States of America, 95, 2979–2984. 10.1073/pnas.95.6.2979 - DOI - PMC - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous

