Dibenzofuranylethylamines as 5-HT2A/2C Receptor Agonists
- PMID: 32064387
- PMCID: PMC7016908
- DOI: 10.1021/acsomega.9b03430
Dibenzofuranylethylamines as 5-HT2A/2C Receptor Agonists
Abstract
The human 5-HT2 receptor subtypes have high sequence identity in their orthosteric ligand-binding domain, and many agonists are poorly selective between the 5-HT2A and 5-HT2C subtypes. Nevertheless, their activation is associated with different pharmacological outcomes. We synthesized five phenethylamine analogs in which the benzene ring is replaced by a bulky dibenzo[b,d]furan moiety and found a couple with >70-fold 5-HT2C selectivity. Molecular docking studies of the most potent compound (5) at both receptor subtypes revealed the likely structural basis of its selectivity. Although in both cases, some crucial interactions are conserved, the change of the Ala2225.46 residue in the 5-HT2C receptor to the larger Ser2425.46 in the 5-HT2A subtype, which is the only structural difference between the orthosteric binding pockets of both receptors, weakens a π-π stacking interaction between the dibenzofuran moiety and the important Phe6.52 residue and breaks a hydrogen bond between the dibenzofuran oxygen and Ser5.43, explaining the selectivity of compound 5 for the 5-HT2C receptor. We believe that this effect of the residue at position 5.46 merits further exploration in the search for selective 5-HT2C receptor agonists that are of considerable interest in the treatment of schizophrenia and substance abuse.
Copyright © 2020 American Chemical Society.
Conflict of interest statement
The authors declare no competing financial interest.
Figures

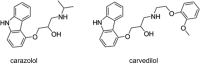
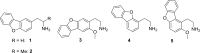
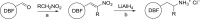



References
-
- Huang X.-P.; Setola V.; Yadav P. N.; Allen J. A.; Rogan S. C.; Hanson B. J.; Revankar C.; Robers M.; Doucette C.; Roth B. L. Parallel functional activity profiling reveals valvulopathogens are potent 5-hydroxytryptamine2Breceptor agonists: Implications for drug safety assessment. Mol. Pharmacol. 2009, 76, 710–722. 10.1124/mol.109.058057. - DOI - PMC - PubMed
-
- Shulgin A. T.; Shulgin A.. PIHKAL, Phenethylamines I Have Known and Loved, Transform Press, Berkeley, CA. 1991.
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous
