Differential kidney proximal tubule cell responses to protein overload by albumin and its ligands
- PMID: 32068462
- PMCID: PMC7099508
- DOI: 10.1152/ajprenal.00490.2019
Differential kidney proximal tubule cell responses to protein overload by albumin and its ligands
Abstract
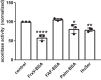
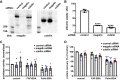
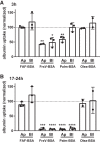
Albuminuria is frequently associated with proximal tubule (PT) cytotoxicity that can feed back to cause glomerular damage and exacerbate kidney disease. PT cells express megalin and cubilin receptors that bind to and internalize albumin over a broad concentration range. How the exposure to high concentrations of albumin leads to PT cytotoxicity remains unclear. Fatty acids and other ligands bound to albumin are known to trigger production of reactive oxygen species (ROS) that impair PT function. Alternatively or in addition, uptake of high concentrations of albumin may overload the endocytic pathway and elicit downstream responses. Here, we used a well-differentiated PT cell culture model with high endocytic capacity to dissect the effects of albumin versus its ligands on endocytic uptake and degradation of albumin, production of ROS, and cell viability. Cellular responses differed dramatically, depending on the preparation of albumin tested. Knockdown of megalin or cubilin failed to prevent ROS production mediated by albumin ligands, suggesting that receptor-mediated internalization of albumin was not necessary to trigger cellular responses to albumin ligands. Moreover, albumin induced cytotoxic responses when added to the basolateral surface of PT cells. Whereas overnight incubation with high concentrations of fatty acid-free albumin had no overt effects on cell function or viability, lysosomal degradation kinetics were slowed upon longer exposure, consistent with overload of the PT endocytic/degradative pathway. Together, the results of our study demonstrate that the PT responds independently to albumin and to its ligands and suggest that the consequences of albumin overload in vivo may be dependent on metabolic state.
Keywords: endocytosis; lipotoxicity; palmitate; proteinuria; proximal tubule; reactive oxygen species.
Conflict of interest statement
No conflicts of interest, financial or otherwise, are declared by the authors.
Figures
Similar articles
-
Receptor-associated protein impairs ligand binding to megalin and megalin-dependent endocytic flux in proximal tubule cells.Am J Physiol Renal Physiol. 2023 Oct 1;325(4):F457-F464. doi: 10.1152/ajprenal.00165.2023. Epub 2023 Aug 3. Am J Physiol Renal Physiol. 2023. PMID: 37534387 Free PMC article.
-
Distinct functions of megalin and cubilin receptors in recovery of normal and nephrotic levels of filtered albumin.Am J Physiol Renal Physiol. 2020 May 1;318(5):F1284-F1294. doi: 10.1152/ajprenal.00030.2020. Epub 2020 Mar 23. Am J Physiol Renal Physiol. 2020. PMID: 32200668 Free PMC article.
-
Cubilin is essential for albumin reabsorption in the renal proximal tubule.J Am Soc Nephrol. 2010 Nov;21(11):1859-67. doi: 10.1681/ASN.2010050492. Epub 2010 Aug 26. J Am Soc Nephrol. 2010. PMID: 20798259 Free PMC article.
-
Chloride channels and endocytosis: new insights from Dent's disease and ClC-5 knockout mice.Nephron Physiol. 2005;99(3):p69-73. doi: 10.1159/000083210. Nephron Physiol. 2005. PMID: 15637424 Review.
-
Albumin uptake and processing by the proximal tubule: physiological, pathological, and therapeutic implications.Physiol Rev. 2022 Oct 1;102(4):1625-1667. doi: 10.1152/physrev.00014.2021. Epub 2022 Apr 4. Physiol Rev. 2022. PMID: 35378997 Free PMC article. Review.
Cited by
-
Albumin is an interface between blood plasma and cell membrane, and not just a sponge.Clin Kidney J. 2021 Oct 5;15(4):624-634. doi: 10.1093/ckj/sfab194. eCollection 2022 Apr. Clin Kidney J. 2021. PMID: 35371452 Free PMC article.
-
TCA Cycle and Fatty Acids Oxidation Reflect Early Cardiorenal Damage in Normoalbuminuric Subjects with Controlled Hypertension.Antioxidants (Basel). 2021 Jul 9;10(7):1100. doi: 10.3390/antiox10071100. Antioxidants (Basel). 2021. PMID: 34356333 Free PMC article.
-
Crosstalk between glomeruli and tubules.Nat Rev Nephrol. 2025 Mar;21(3):189-199. doi: 10.1038/s41581-024-00907-0. Epub 2024 Dec 6. Nat Rev Nephrol. 2025. PMID: 39643696 Review.
-
TRPC6 Inactivation Reduces Albuminuria Induced by Protein Overload in Sprague Dawley Rats.Cells. 2022 Jun 21;11(13):1985. doi: 10.3390/cells11131985. Cells. 2022. PMID: 35805070 Free PMC article.
-
Evidence of chronic kidney injury in patients not meeting KDIGO criteria for chronic kidney disease.Clin Kidney J. 2022 Jan 12;15(7):1217-1220. doi: 10.1093/ckj/sfac007. eCollection 2022 Jul. Clin Kidney J. 2022. PMID: 35756746 Free PMC article.
References
-
- Dixon R, Brunskill NJ. Activation of mitogenic pathways by albumin in kidney proximal tubule epithelial cells: implications for the pathophysiology of proteinuric states. J Am Soc Nephrol 10: 1487–1497, 1999. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

