Phasic signaling in the bed nucleus of the stria terminalis during fear learning predicts within- and across-session cued fear expression
- PMID: 32071254
- PMCID: PMC7029722
- DOI: 10.1101/lm.050807.119
Phasic signaling in the bed nucleus of the stria terminalis during fear learning predicts within- and across-session cued fear expression
Abstract
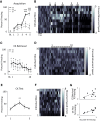
While results from many past studies have implicated the bed nucleus of the stria terminalis (BNST) in mediating the expression of sustained negative affect, recent studies have highlighted a more complex role for BNST that includes aspects of fear learning in addition to defensive responding. As BNST is thought to encode ambiguous or unpredictable threat, it seems plausible that it may be involved in encoding early cued fear learning, especially immediately following a first tone-shock pairing when the conditioned stimulus-unconditioned stimulus (CS-US) contingency is not fully apparent. To investigate this, we conducted in vivo electrophysiological recording studies to examine neural dynamics of BNST units during cued fear acquisition and recall. We identified two functionally distinct subpopulations of BNST neurons that encode the intertrial interval (ITI) and may contribute to within- and across-session fear learning. "Ramping" cell activity during cued fear acquisition parallels the increase in freezing expression as mice learn the CS-US contingency, while "Phasic" cells encode postshock (USpost) periods (30 sec following encounter with footshock) only during early trials. Importantly, the magnitude of Phasic unit responsivity to the first USpost period predicted not only freezing expression in response to the subsequent CS during acquisition, but also CS freezing evoked 24 h later during CS retrieval. These findings suggest for the first time that BNST activity may serve as an instructive signal during cued fear learning.
© 2020 Bjorni et al.; Published by Cold Spring Harbor Laboratory Press.
Figures

References
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous
