Sucrose promotes caries progression by disrupting the microecological balance in oral biofilms: an in vitro study
- PMID: 32076013
- PMCID: PMC7031525
- DOI: 10.1038/s41598-020-59733-6
Sucrose promotes caries progression by disrupting the microecological balance in oral biofilms: an in vitro study
Abstract
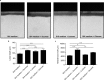
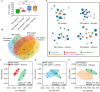
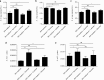
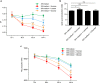
Sucrose has long been regarded as the most cariogenic carbohydrate. However, why sucrose causes severer dental caries than other sugars is largely unknown. Considering that caries is a polymicrobial infection resulting from dysbiosis of oral biofilms, we hypothesized that sucrose can introduce a microbiota imbalance favoring caries to a greater degree than other sugars. To test this hypothesis, an in vitro saliva-derived multispecies biofilm model was established, and by comparing caries lesions on enamel blocks cocultured with biofilms treated with sucrose, glucose and lactose, we confirmed that this model can reproduce the in vivo finding that sucrose has the strongest cariogenic potential. In parallel, compared to a control treatment, sucrose treatment led to significant changes within the microbial structure and assembly of oral microflora, while no significant difference was detected between the lactose/glucose treatment group and the control. Specifically, sucrose supplementation disrupted the homeostasis between acid-producing and alkali-producing bacteria. Consistent with microbial dysbiosis, we observed the most significant disequilibrium between acid and alkali metabolism in sucrose-treated biofilms. Taken together, our data indicate that the cariogenicity of sugars is closely related to their ability to regulate the oral microecology. These findings advance our understanding of caries etiology from an ecological perspective.
Conflict of interest statement
The authors declare no competing interests.
Figures


References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical

