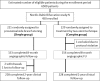
Randomised comparison of provisional side branch stenting versus a two-stent strategy for treatment of true coronary bifurcation lesions involving a large side branch: the Nordic-Baltic Bifurcation Study IV
- PMID: 32076558
- PMCID: PMC6999681
- DOI: 10.1136/openhrt-2018-000947
Randomised comparison of provisional side branch stenting versus a two-stent strategy for treatment of true coronary bifurcation lesions involving a large side branch: the Nordic-Baltic Bifurcation Study IV
Abstract
Background: It is still uncertain whether coronary bifurcations with lesions involving a large side branch (SB) should be treated by stenting the main vessel and provisional stenting of the SB (simple) or by routine two-stent techniques (complex). We aimed to compare clinical outcome after treatment of lesions in large bifurcations by simple or complex stent implantation.
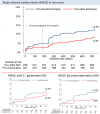
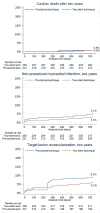
Methods: The study was a randomised, superiority trial. Enrolment required a SB≥2.75 mm, ≥50% diameter stenosis in both vessels, and allowed SB lesion length up to 15 mm. The primary endpoint was a composite of cardiac death, non-procedural myocardial infarction and target lesion revascularisation at 6 months. Two-year clinical follow-up was included in this primary reporting due to lower than expected event rates.
Results: A total of 450 patients were assigned to simple stenting (n=221) or complex stenting (n=229) in 14 Nordic and Baltic centres. Two-year follow-up was available in 218 (98.6%) and 228 (99.5%) patients, respectively. The primary endpoint of major adverse cardiac events (MACE) at 6 months was 5.5% vs 2.2% (risk differences 3.2%, 95% CI -0.2 to 6.8, p=0.07) and at 2 years 12.9% vs 8.4% (HR 0.63, 95% CI 0.35 to 1.13, p=0.12) after simple versus complex treatment. In the subgroup treated by newer generation drug-eluting stents, MACE was 12.0% vs 5.6% (HR 0.45, 95% CI 0.17 to 1.17, p=0.10) after simple versus complex treatment.
Conclusion: In the treatment of bifurcation lesions involving a large SB with ostial stenosis, routine two-stent techniques did not improve outcome significantly compared with treatment by the simpler main vessel stenting technique after 2 years.
Trial registration number: NCT01496638.
Keywords: complex coronary lesions; coronary bifurcations; drug eluting stents.
© Author(s) (or their employer(s)) 2020. Re-use permitted under CC BY-NC. No commercial re-use. See rights and permissions. Published by BMJ.
Conflict of interest statement
Competing interests: IK received institutional research grants from Cordis, Abbott and speaker fee from Astra Zeneca. NRH has received institutional research grants from Cordis, Abbott, Terumo, Biosensors, Biotronik, Medis medical imaging, Reva Medical, Boston Scientific, St. Jude Medical and Medtronic and has received speaker fees and personal honorarium from St. Jude Medical, Terumo, Reva Medical and Biotronik. LOJ has received institutional research grants from Terumo, Biosensors and Biotronik. JFL has received institutional research grants from Cordis, Abbott, Terumo, Biosensors, Biotronik, Medis medical imaging, Reva Medical, Boston Scientific, Heartflow, St. Jude Medical and Medtronic and has received speaker fees from Biotronik, Biosensors, Tryton, Boston Scientific, St. Jude Medical, Terumo, Reva medical, Cordis, Astra Zeneca and Abbott. GL has received speaker fees from Astra Zeneca.
Figures

References
Publication types
MeSH terms
Associated data
LinkOut - more resources
Full Text Sources
Medical
