Optimized functional and structural design of dual-target LMRAP, a bifunctional fusion protein with a 25-amino-acid antitumor peptide and GnRH Fc fragment
- PMID: 32082972
- PMCID: PMC7016293
- DOI: 10.1016/j.apsb.2019.10.010
Optimized functional and structural design of dual-target LMRAP, a bifunctional fusion protein with a 25-amino-acid antitumor peptide and GnRH Fc fragment
Abstract
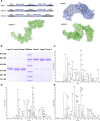
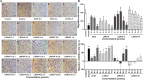
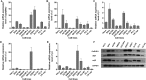
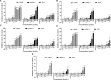
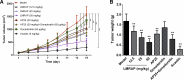
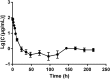
To develop fusion protein of a GnRH Fc fragment and the integrin targeting AP25 antitumor peptide for GnRH receptor-expressing cancer therapy. The LMRAP fusion protein was constructed. A transwell invasion assay was performed. The gene mRNA and protein levels of GnRHR-I, α5β1, and αvβ3 in different cancer cell lines were assessed. Cell proliferation was measured using a cell counting kit-8. An antagonist assay was performed on GnRH receptors. Anti-tumor activity was evaluated with a mouse xenograft tumor model. Immunohistochemistry (IHC) was applied to detect CD31 and CD34 expressions. Pharmacokinetic characteristics were determined with an indirect competition ELISA. The developed bifunctional fusion protein LMRAP not only inhibited HUVEC invasion, but also inhibited proliferation of GnRHR-I, α5β1, and αvβ3 high expression cancer cells. The IC50 for LMRAP in the GnRH receptor was 6.235 × 10-4 mol/L. LMRAP significantly inhibited human prostate cancer cell line 22RV1 proliferation in vivo and in vitro. LMRAP significantly inhibited CD31 and CD34 expressions. The elimination half-life of the fusion protein LMRAP was 33 h in rats. The fusion protein made of a GnRH Fc fragment and the integrin targeting AP25 peptide retained the bifunctional biological activity of GnRHR blocking, angiogenesis inhibition, prolonged half-life and good tolerance.
Keywords: Angiogenesis; Fusion protein; GnRH; Integrin; Prostate cancer.
© 2020 Chinese Pharmaceutical Association and Institute of Materia Medica, Chinese Academy of Medical Sciences. Production and hosting by Elsevier B.V.
Figures



References
-
- Stanislaus D., Pinter J.H., Janovick J.A., Conn P.M. Mechanisms mediating multiple physiological responses to gonadotropin-releasing hormone. Mol Cell Endocrinol. 1998;144:1–10. - PubMed
-
- Bono A.V., Salvadore M., Celato N. Gonadotropin-releasing hormone receptors in prostate tissue. Anal Quant Cytol Histol. 2002;24:221–227. - PubMed
-
- Halmos G., Arencibia J.M., Schally A.V., Davis R., Bostwick D.G. High incidence of receptors for luteinizing hormone-releasing hormone (LHRH) and LHRH receptor gene expression in human prostate cancers. J Urol. 2000;163:623–629. - PubMed
-
- Imai A., Tamaya T. GnRH receptor and apoptotic signaling. Vitam Horm. 2000;59:1–33. - PubMed
-
- Emons G., Grundker C., Gunthert A.R., Westphalen S., Kavanagh J., Verschraegen C. GnRH antagonists in the treatment of gynecological and breast cancers. Endocr Relat Cancer. 2003;10:291–299. - PubMed
LinkOut - more resources
Full Text Sources

