Statin-mediated cholesterol depletion exerts coordinated effects on the alterations in rat vascular smooth muscle cell biomechanics and migration
- PMID: 32083311
- PMCID: PMC7394245
- DOI: 10.1113/JP279528
Statin-mediated cholesterol depletion exerts coordinated effects on the alterations in rat vascular smooth muscle cell biomechanics and migration
Abstract
Key points: This study demonstrates and evaluates the changes in rat vascular smooth muscle cell biomechanics following statin-mediated cholesterol depletion. Evidence is presented to show correlated changes in migration and adhesion of vascular smooth muscle cells to extracellular matrix proteins fibronectin and collagen. Concurrently, integrin α5 expression was enhanced but not integrin α2. Atomic force microscopy analysis provides compelling evidence of coordinated reduction in vascular smooth muscle cell stiffness and actin cytoskeletal orientation in response to statin-mediated cholesterol depletion. Proof is provided that statin-mediated cholesterol depletion remodels total vascular smooth muscle cell cytoskeletal orientation that may additionally participate in altering ex vivo aortic vessel function. It is concluded that statin-mediated cholesterol depletion may coordinate vascular smooth muscle cell migration and adhesion to different extracellular matrix proteins and regulate cellular stiffness and cytoskeletal orientation, thus impacting the biomechanics of the cell.
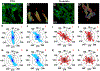
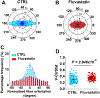
Abstract: Not only does cholesterol induce an inflammatory response and deposits in foam cells at the atherosclerotic plaque, it also regulates cellular mechanics, proliferation and migration in atherosclerosis progression. Statins are HMG-CoA reductase inhibitors that are known to inhibit cellular cholesterol biosynthesis and are clinically prescribed to patients with hypercholesterolemia or related cardiovascular conditions. Nonetheless, the effect of statin-mediated cholesterol management on cellular biomechanics is not fully understood. In this study, we aimed to assess the effect of fluvastatin-mediated cholesterol management on primary rat vascular smooth muscle cell (VSMC) biomechanics. Real-time measurement of cell adhesion, stiffness, and imaging were performed using atomic force microscopy (AFM). Cellular migration on extra cellular matrix (ECM) protein surfaces was studied by time-lapse imaging. The effect of changes in VSMC biomechanics on aortic function was assessed using an ex vivo myograph system. Fluvastatin-mediated cholesterol depletion (-27.8%) lowered VSMC migration distance on a fibronectin (FN)-coated surface (-14.8%) but not on a type 1 collagen (COL1)-coated surface. VSMC adhesion force to FN (+33%) and integrin α5 expression were enhanced but COL1 adhesion and integrin α2 expression were unchanged upon cholesterol depletion. In addition, VSMC stiffness (-46.6%) and ex vivo aortic ring contraction force (-40.1%) were lowered and VSMC actin cytoskeletal orientation was reduced (-24.5%) following statin-mediated cholesterol depletion. Altogether, it is concluded that statin-mediated cholesterol depletion may coordinate VSMC migration and adhesion to different ECM proteins and regulate cellular stiffness and cytoskeletal orientation, thus impacting the biomechanics of the cell and aortic function.
Keywords: atomic force microscopy; cell adhesion; cell mechanics; cholesterol; cytoskeleton; fluvastatin; vascular smooth muscle cells.
© 2020 The Authors. The Journal of Physiology © 2020 The Physiological Society.
Conflict of interest statement
Competing intrests
The authors declare that they have no competing interests.
Figures






Comment in
-
Impact of statins on vascular smooth muscle cells and relevance to atherosclerosis.J Physiol. 2020 Jun;598(12):2295-2296. doi: 10.1113/JP279774. Epub 2020 May 18. J Physiol. 2020. PMID: 32329895 No abstract available.
References
-
- Allahverdian S, Chehroudi AC, McManus BM, Abraham T & Francis GA. (2014). Contribution of intimal smooth muscle cells to cholesterol accumulation and macrophage-like cells in human atherosclerosis. Circulation 129, 1551–1559. - PubMed
-
- Allahverdian S, Pannu PS & Francis GA. (2012). Contribution of monocyte-derived macrophages and smooth muscle cells to arterial foam cell formation. Cardiovasc Res 95, 165–172. - PubMed
-
- Ayee MA & Levitan I. (2016). Paradoxical impact of cholesterol on lipid packing and cell stiffness. Front Biosci (Landmark Ed) 21, 1245–1259. - PubMed
-
- Basatemur GL, Jorgensen HF, Clarke MCH, Bennett MR & Mallat Z. (2019). Vascular smooth muscle cells in atherosclerosis. Nat Rev Cardiol. - PubMed
-
- Bellosta S, Arnaboldi L, Gerosa L, Canavesi M, Parente R, Baetta R, Paoletti R & Corsini A. (2004). Statins effect on smooth muscle cell proliferation. Semin Vasc Med 4, 347–356. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous

