Effect of Diphtheria Toxin-Based Gene Therapy for Hepatocellular Carcinoma
- PMID: 32085552
- PMCID: PMC7072394
- DOI: 10.3390/cancers12020472
Effect of Diphtheria Toxin-Based Gene Therapy for Hepatocellular Carcinoma
Abstract
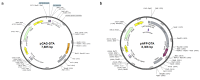
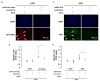
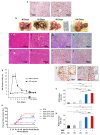
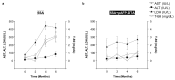
Hepatocellular carcinoma (HCC) is a major global malignancy, responsible for >90% of primary liver cancers. Currently available therapeutic options have poor performances due to the highly heterogeneous nature of the tumor cells; recurrence is highly probable, and some patients develop resistances to the therapies. Accordingly, the development of a novel therapy is essential. We assessed gene therapy for HCC using a diphtheria toxin fragment A (DTA) gene-expressing plasmid, utilizing a non-viral hydrodynamics-based procedure. The antitumor effect of DTA expression in HCC cell lines (and alpha-fetoprotein (AFP) promoter selectivity) is assessed in vitro by examining HCC cell growth. Moreover, the effect and safety of the AFP promoter-selective DTA expression was examined in vivo using an HCC mice model established by the hydrodynamic gene delivery of the yes-associated protein (YAP)-expressing plasmid. The protein synthesis in DTA transfected cells is inhibited by the disappearance of tdTomato and GFP expression co-transfected upon the delivery of the DTA plasmid; the HCC cell growth is inhibited by the expression of DTA in HCC cells in an AFP promoter-selective manner. A significant inhibition of HCC occurrence and the suppression of the tumor marker of AFP and des-gamma-carboxy prothrombin can be seen in mice groups treated with hydrodynamic gene delivery of DTA, both 0 and 2 months after the YAP gene delivery. These results suggest that DTA gene therapy is effective for HCC.
Keywords: alpha-fetoprotein; diphtheria toxin fragment A; gene therapy; hepatocellular carcinoma; hydrodynamic gene delivery.
Conflict of interest statement
The authors declare that they have no current financial arrangement or affiliation with any organization that may have a direct influence on their work.
Figures



References
-
- European Association for the Study of the Liver European association for the study of the liver EASL clinical practice guidelines: management of hepatocellular carcinoma. J. Hepatol. 2018;69:182–236. - PubMed
Grants and funding
LinkOut - more resources
Full Text Sources

