Comparative Interactome Analysis of Emerin, MAN1 and LEM2 Reveals a Unique Role for LEM2 in Nucleotide Excision Repair
- PMID: 32085595
- PMCID: PMC7072835
- DOI: 10.3390/cells9020463
Comparative Interactome Analysis of Emerin, MAN1 and LEM2 Reveals a Unique Role for LEM2 in Nucleotide Excision Repair
Abstract
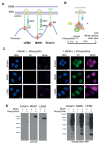
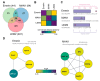
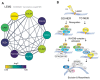
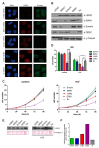
LAP2-Emerin-MAN1 (LEM) domain-containing proteins represent an abundant group of inner nuclear membrane proteins involved in diverse nuclear functions, but their functional redundancies remain unclear. Here, using the biotinylation-dependent proximity approach, we report proteome-wide comparative interactome analysis of the two structurally related LEM proteins MAN1 (LEMD3) and LEM2 (LEMD2), and the more distantly related emerin (EMD). While over 60% of the relatively small group of MAN1 and emerin interactors were also found in the LEM2 interactome, the latter included a large number of candidates (>85%) unique for LEM2. The interacting partners unique for emerin support and provide further insight into the previously reported role of emerin in centrosome positioning, and the MAN1-specific interactors suggest a role of MAN1 in ribonucleoprotein complex assembly. Interestingly, the LEM2-specific interactome contained several proteins of the nucleotide excision repair pathway. Accordingly, LEM2-depleted cells, but not MAN1- and emerin-depleted cells, showed impaired proliferation following ultraviolet-C (UV-C) irradiation and prolonged accumulation of γH2AX, similar to cells deficient in the nucleotide excision repair protein DNA damage-binding protein 1 (DDB1). These findings indicate impaired DNA damage repair in LEM2-depleted cells. Overall, this interactome study identifies new potential interaction partners of emerin, MAN1 and particularly LEM2, and describes a novel potential involvement of LEM2 in nucleotide excision repair at the nuclear periphery.
Keywords: BioID; DNA repair; LEM-proteins; inner nuclear membrane; nuclear envelope; nucleotide excision repair.
Conflict of interest statement
The funders had no role in the design of the study; in the collection, analyses, or interpretation of data; in the writing of the manuscript, or in the decision to publish the results.
Figures
Similar articles
-
LEM2 is a novel MAN1-related inner nuclear membrane protein associated with A-type lamins.J Cell Sci. 2005 Dec 15;118(Pt 24):5797-810. doi: 10.1242/jcs.02701. J Cell Sci. 2005. PMID: 16339967
-
Direct binding of nuclear membrane protein MAN1 to emerin in vitro and two modes of binding to barrier-to-autointegration factor.J Biol Chem. 2005 Apr 8;280(14):13863-70. doi: 10.1074/jbc.M413020200. Epub 2005 Jan 29. J Biol Chem. 2005. PMID: 15681850
-
Overlapping functions of nuclear envelope proteins NET25 (Lem2) and emerin in regulation of extracellular signal-regulated kinase signaling in myoblast differentiation.Mol Cell Biol. 2009 Nov;29(21):5718-28. doi: 10.1128/MCB.00270-09. Epub 2009 Aug 31. Mol Cell Biol. 2009. PMID: 19720741 Free PMC article.
-
Beyond Tethering and the LEM domain: MSCellaneous functions of the inner nuclear membrane Lem2.Nucleus. 2016 Nov;7(6):523-531. doi: 10.1080/19491034.2016.1252892. Epub 2016 Oct 31. Nucleus. 2016. PMID: 27797637 Free PMC article. Review.
-
All in the family: evidence for four new LEM-domain proteins Lem2 (NET-25), Lem3, Lem4 and Lem5 in the human genome.Symp Soc Exp Biol. 2004;(56):329-39. Symp Soc Exp Biol. 2004. PMID: 15565891 Review.
Cited by
-
Emerin Phosphorylation during the Early Phase of the Oxidative Stress Response Influences Emerin-BAF Interaction and BAF Nuclear Localization.Cells. 2020 Jun 6;9(6):1415. doi: 10.3390/cells9061415. Cells. 2020. PMID: 32517247 Free PMC article.
-
Shared and Distinctive Neighborhoods of Emerin and Lamin B Receptor Revealed by Proximity Labeling and Quantitative Proteomics.J Proteome Res. 2022 Sep 2;21(9):2197-2210. doi: 10.1021/acs.jproteome.2c00281. Epub 2022 Aug 16. J Proteome Res. 2022. PMID: 35972904 Free PMC article.
-
Nuclear Dynamics and Chromatin Structure: Implications for Pancreatic Cancer.Cells. 2021 Oct 1;10(10):2624. doi: 10.3390/cells10102624. Cells. 2021. PMID: 34685604 Free PMC article. Review.
-
The Diverse Cellular Functions of Inner Nuclear Membrane Proteins.Cold Spring Harb Perspect Biol. 2021 Sep 1;13(9):a040477. doi: 10.1101/cshperspect.a040477. Cold Spring Harb Perspect Biol. 2021. PMID: 33753404 Free PMC article. Review.
-
A novel role for CSA in the regulation of nuclear envelope integrity: uncovering a non-canonical function.Life Sci Alliance. 2024 Aug 29;7(11):e202402745. doi: 10.26508/lsa.202402745. Print 2024 Nov. Life Sci Alliance. 2024. PMID: 39209536 Free PMC article.
References
-
- Cheng L.C., Baboo S., Lindsay C., Brusman L., Martinez-Bartolome S., Tapia O., Zhang X., Yates J.R., 3rd, Gerace L. Identification of new transmembrane proteins concentrated at the nuclear envelope using organellar proteomics of mesenchymal cells. Nucleus. 2019;10:126–143. doi: 10.1080/19491034.2019.1618175. - DOI - PMC - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

