Localization of Annexin A6 in Matrix Vesicles During Physiological Mineralization
- PMID: 32085611
- PMCID: PMC7072960
- DOI: 10.3390/ijms21041367
Localization of Annexin A6 in Matrix Vesicles During Physiological Mineralization
Abstract
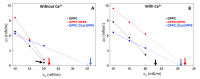
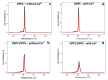
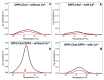
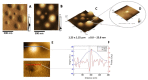
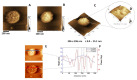
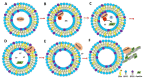
Annexin A6 (AnxA6) is the largest member of the annexin family of proteins present in matrix vesicles (MVs). MVs are a special class of extracellular vesicles that serve as a nucleation site during cartilage, bone, and mantle dentin mineralization. In this study, we assessed the localization of AnxA6 in the MV membrane bilayer using native MVs and MV biomimetics. Biochemical analyses revealed that AnxA6 in MVs can be divided into three distinct groups. The first group corresponds to Ca2+-bound AnxA6 interacting with the inner leaflet of the MV membrane. The second group corresponds to AnxA6 localized on the surface of the outer leaflet. The third group corresponds to AnxA6 inserted in the membrane's hydrophobic bilayer and co-localized with cholesterol (Chol). Using monolayers and proteoliposomes composed of either dipalmitoylphosphatidylcholine (DPPC) to mimic the outer leaflet of the MV membrane bilayer or a 9:1 DPPC:dipalmitoylphosphatidylserine (DPPS) mixture to mimic the inner leaflet, with and without Ca2+, we confirmed that, in agreement with the biochemical data, AnxA6 interacted differently with the MV membrane. Thermodynamic analyses based on the measurement of surface pressure exclusion (πexc), enthalpy (ΔH), and phase transition cooperativity (Δt1/2) showed that AnxA6 interacted with DPPC and 9:1 DPPC:DPPS systems and that this interaction increased in the presence of Chol. The selective recruitment of AnxA6 by Chol was observed in MVs as probed by the addition of methyl-β-cyclodextrin (MβCD). AnxA6-lipid interaction was also Ca2+-dependent, as evidenced by the increase in πexc in negatively charged 9:1 DPPC:DPPS monolayers and the decrease in ΔH in 9:1 DPPC:DPPS proteoliposomes caused by the addition of AnxA6 in the presence of Ca2+ compared to DPPC zwitterionic bilayers. The interaction of AnxA6 with DPPC and 9:1 DPPC:DPPS systems was distinct even in the absence of Ca2+ as observed by the larger change in Δt1/2 in 9:1 DPPC:DPPS vesicles as compared to DPPC vesicles. Protrusions on the surface of DPPC proteoliposomes observed by atomic force microscopy suggested that oligomeric AnxA6 interacted with the vesicle membrane. Further work is needed to delineate possible functions of AnxA6 at its different localizations and ways of interaction with lipids.
Keywords: Annexin A6; Langmuir monolayers; biomineralization; differential scanning calorimetry.; matrix vesicles; proteoliposomes.
Conflict of interest statement
The authors declare no conflict of interest.
Figures


References
-
- Wu L.N., Genge B.R., Lloyd G.C., Wuthier R.E. Collagen-binding proteins in collagenase-released matrix vesicles from cartilage. Interaction between matrix vesicle proteins and different types of collagen. J. Biol. Chem. 1991;266:1195–1203. - PubMed
-
- Kirsch T., Wuthier R.E. Stimulation of calcification of growth plate cartilage matrix vesicles by binding to type II and X collagens. J. Biol. Chem. 1994;269:11462–11469. - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

