A novel postsynaptic signal pathway of sympathetic neural regulation of murine colonic motility
- PMID: 32086857
- PMCID: PMC7147814
- DOI: 10.1096/fj.201903134R
A novel postsynaptic signal pathway of sympathetic neural regulation of murine colonic motility
Abstract
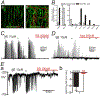
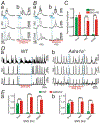
Transcriptome data revealed α1 adrenoceptors (ARs) expression in platelet-derived growth factor receptor α+ cells (PDGFRα+ cells) in murine colonic musculature. The role of PDGFRα+ cells in sympathetic neural regulation of murine colonic motility was investigated. Norepinephrine (NE), via α1A ARs, activated a small conductance Ca2+ -activated K+ (SK) conductance, evoked outward currents and hyperpolarized PDGFRα+ cells (the α1A AR-SK channel signal pathway). α1 AR agonists increased intracellular Ca2+ transients in PDGFRα+ cells and inhibited spontaneous phasic contractions (SPCs) of colonic muscle through activation of a SK conductance. Sympathetic nerve stimulation inhibited both contractions of distal colon and propulsive contractions represented by the colonic migrating motor complexes (CMMCs) via the α1A AR-SK channel signal pathway. Postsynaptic signaling through α1A ARs in PDGFRα+ cells is a novel mechanism that conveys part of stress responses in the colon. PDGFRα+ cells appear to be a primary effector of sympathetic neural regulation of murine colonic motility.
Keywords: PDGFRα+ cells; colonic motility; sympathetic neural regulation; α1 adrenoceptor.
© 2020 Federation of American Societies for Experimental Biology.
Figures





Similar articles
-
Norepinephrine Has Dual Effects on Human Colonic Contractions Through Distinct Subtypes of Alpha 1 Adrenoceptors.Cell Mol Gastroenterol Hepatol. 2020;10(3):658-671.e1. doi: 10.1016/j.jcmgh.2020.04.015. Epub 2020 May 4. Cell Mol Gastroenterol Hepatol. 2020. PMID: 32376421 Free PMC article.
-
Purinergic inhibitory regulation of murine detrusor muscles mediated by PDGFRα+ interstitial cells.J Physiol. 2014 Mar 15;592(6):1283-93. doi: 10.1113/jphysiol.2013.267989. Epub 2014 Jan 6. J Physiol. 2014. PMID: 24396055 Free PMC article.
-
Platelet-derived growth factor receptor-α-positive cells and not smooth muscle cells mediate purinergic hyperpolarization in murine colonic muscles.Am J Physiol Cell Physiol. 2014 Sep 15;307(6):C561-70. doi: 10.1152/ajpcell.00080.2014. Epub 2014 Jul 23. Am J Physiol Cell Physiol. 2014. PMID: 25055825 Free PMC article.
-
Sympathetically evoked Ca2+ signaling in arterial smooth muscle.Acta Pharmacol Sin. 2006 Dec;27(12):1515-25. doi: 10.1111/j.1745-7254.2006.00465.x. Acta Pharmacol Sin. 2006. PMID: 17112404 Review.
-
The pharmacology of α1-adrenoceptor subtypes.Eur J Pharmacol. 2019 Jul 15;855:305-320. doi: 10.1016/j.ejphar.2019.04.047. Epub 2019 May 5. Eur J Pharmacol. 2019. PMID: 31067439 Review.
Cited by
-
Circadian rhythms in colonic function.Front Physiol. 2023 Aug 30;14:1239278. doi: 10.3389/fphys.2023.1239278. eCollection 2023. Front Physiol. 2023. PMID: 37711458 Free PMC article. Review.
-
Ca2+ dynamics in interstitial cells: foundational mechanisms for the motor patterns in the gastrointestinal tract.Physiol Rev. 2024 Jan 1;104(1):329-398. doi: 10.1152/physrev.00036.2022. Epub 2023 Aug 10. Physiol Rev. 2024. PMID: 37561138 Free PMC article. Review.
-
Pharmacological characterization of alpha adrenoceptor-mediated motor responses in the rat colon.Neurogastroenterol Motil. 2025 Jan;37(1):e14921. doi: 10.1111/nmo.14921. Epub 2024 Sep 30. Neurogastroenterol Motil. 2025. PMID: 39344996 Free PMC article.
-
Immunohistochemical characterization of interstitial cells and their spatial relationship to motor neurons within the mouse esophagus.Cell Tissue Res. 2025 Jan;399(1):61-84. doi: 10.1007/s00441-024-03929-z. Epub 2024 Nov 28. Cell Tissue Res. 2025. PMID: 39607495
-
Neural targets of the enteric dopaminergic system in regulating motility of rat proximal colon.Pflugers Arch. 2023 Nov;475(11):1315-1327. doi: 10.1007/s00424-023-02849-1. Epub 2023 Aug 17. Pflugers Arch. 2023. PMID: 37589734
References
-
- Norberg KA and Sjoqvist F (1966) New possibilities for adrenergic modulation of ganglionic transmission. Permacol. Rev 18, 743–751 - PubMed
-
- Burnstock G and Costa M (1973) Inhibitory innervation of the gut. Gastroenterology 64, 141–144 - PubMed
-
- De Ponti F, Giaroni C, Cosentino M, Lecchini S and Frigo G (1996) Adrenergic mechanisms in the control of gastrointestinal motility: from basic science to clinical applications. Pharmacol. Ther 69, 59–78 - PubMed
-
- Lomax AE, Sharkey KA and Furness JB (2010) The participation of the sympathetic innervation of the gastrointestinal tract in disease states. Neurogastroenterol. Motil 22, 7–18 - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Research Materials
Miscellaneous

