Prolonged Progression-Free Survival in a Patient With Malignant Pleural Mesothelioma Following Korean Herbal Medicine Treatment Alone: A Case Report
- PMID: 32100581
- PMCID: PMC7045291
- DOI: 10.1177/1534735420908345
Prolonged Progression-Free Survival in a Patient With Malignant Pleural Mesothelioma Following Korean Herbal Medicine Treatment Alone: A Case Report
Abstract
Korean herbal medicine treatment (KHMT) involves treating with a combination of natural products, which have been used for thousands of years. Recently, it has been reported to be effective and safe in cancer patients. This case report demonstrates the efficacy of KHMT in a 49-year-old man with malignant pleural mesothelioma (MPM), a rare and highly aggressive cancer. The patient showed recurrent pleural effusion and was diagnosed with epithelioid MPM at cT3NxM0 stage III in December 2017. The multidisciplinary care team recommended multimodal treatment based on an extrapleural pneumonectomy, but he refused this because the treatment was aggressive and the effectiveness was unclear. He decided to undergo pemetrexed plus cisplatin chemotherapy if his condition worsened. He visited the Korean Medicine Cancer Center for alternative treatment options. A KHMT regimen, consisting of twice-daily Gunchil-dan and thrice-daily Bangam-tang, was initiated in December 2017. Since commencement of KHMT, computed tomography and X-ray imaging scans have shown no significant interval changes and progression. At 21 months into treatment (September 2019), no significant adverse events have occurred. Given that the median overall survival of patients with MPM is approximately 1 year, the ongoing progression-free survival of this patient for 21 months is relatively long. This case, therefore, suggests that KHMT is a potential treatment option for MPM patients.
Keywords: Korean medicine; complementary and alternative medicine; herbal medicine; malignant pleural mesothelioma; rare cancer.
Conflict of interest statement
Figures
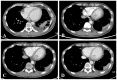
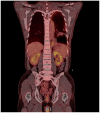
References
-
- American Cancer Society. Cancer facts & figures 2017: special section—rare cancers in adults. https://tinyurl.com/yb4joe3c. Accessed September 10, 2019.
-
- Vogelzang NJ, Rusthoven JJ, Symanowski J, et al. Phase III study of pemetrexed in combination with cisplatin versus cisplatin alone in patients with malignant pleural mesothelioma. J Clin Oncol. 2003;21:2636-2644. - PubMed
-
- Nelson DB, Rice DC, Mitchell KG, et al. Return to intended oncologic treatment after surgery for malignant pleural mesothelioma. J Thorac Cardiovasc Surg. 2019;158:924-929. - PubMed
-
- Yoon SW, Jeong JS, Kim JH, Aggarwal BB. Cancer prevention and therapy: integrating traditional Korean medicine into modern cancer care. Integr Cancer Ther. 2014;13:310-331. - PubMed
-
- US Department of Health and Human Services; National Institutes of Health; National Cancer Institute. Common Terminology Criteria for Adverse Events (CTCAE), Version 5.0. https://ctep.cancer.gov/protocoldevelopment/electronic_applications/docs.... Published November 27, 2017. Accessed September 10, 2019.
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous

