In vitro evaluations on canine monocyte-derived dendritic cells of a nanoparticles delivery system for vaccine antigen against Echinococcus granulosus
- PMID: 32101539
- PMCID: PMC7043750
- DOI: 10.1371/journal.pone.0229121
In vitro evaluations on canine monocyte-derived dendritic cells of a nanoparticles delivery system for vaccine antigen against Echinococcus granulosus
Abstract
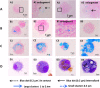
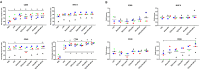
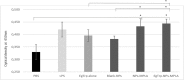
Since dogs play a central role in the contamination of humans and livestock with Echinococcus granulosus, the development of an effective vaccine for dogs is essential to control the disease caused by this parasite. For this purpose, a formulation based on biodegradable polymeric nanoparticles (NPs) as delivery system of recombinant Echinococcus granulosus antigen (tropomyosin EgTrp) adjuved with monophosphoryl lipid A (MPLA) has been developed. The obtained nanoparticles had a size of approximately 200 nm in diameter into which the antigen was correctly preserved and encapsulated. The efficiency of this system to deliver the antigen was evaluated in vitro on canine monocyte-derived dendritic cells (cMoDCs) generated from peripheral blood monocytes. After 48 h of contact between the formulations and cMoDCs, we observed no toxic effect on the cells but a strong internalization of the NPs, probably through different pathways depending on the presence or not of MPLA. An evaluation of cMoDCs activation by flow cytometry showed a stronger expression of CD80, CD86, CD40 and MHCII by cells treated with any of the tested formulations or with LPS (positive control) in comparison to cells treated with PBS (negative control). A higher activation was observed for cells challenged with EgTrp-NPs-MPLA compared to EgTrp alone. Formulations with MPLA, even at low ratio of MPLA, give better results than formulations without MPLA, proving the importance of the adjuvant in the nanoparticles structure. Moreover, autologous T CD4+ cell proliferation observed in presence of cMoDCs challenged with EgTrp-NPs-MPLA was higher than those observed after challenged with EgTrp alone (p<0.05). These first results suggest that our formulation could be used as an antigen delivery system to targeting canine dendritic cells in the course of Echinococcus granulosus vaccine development.
Conflict of interest statement
The authors have declared that no competing interests exist.
Figures


References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Research Materials

