Herpes simplex virus type 1 inflammasome activation in proinflammatory human macrophages is dependent on NLRP3, ASC, and caspase-1
- PMID: 32101570
- PMCID: PMC7043765
- DOI: 10.1371/journal.pone.0229570
Herpes simplex virus type 1 inflammasome activation in proinflammatory human macrophages is dependent on NLRP3, ASC, and caspase-1
Abstract
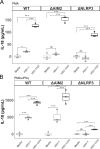
The proinflammatory cytokines interleukin (IL)-1β and IL-18 are products of activation of the inflammasome, an innate sensing system, and important in the pathogenesis of herpes simplex virus type 1 (HSV-1). The release of IL-18 and IL-1β from monocytes/macrophages is critical for protection from HSV-1 based on animal models of encephalitis and genital infection, yet if and how HSV-1 activates inflammasomes in human macrophages is unknown. To investigate this, we utilized both primary human monocyte derived macrophages and human monocytic cell lines (THP-1 cells) with various inflammasome components knocked-out. We found that HSV-1 activates inflammasome signaling in proinflammatory primary human macrophages, but not in resting macrophages. Additionally, HSV-1 inflammasome activation in THP-1 cells is dependent on nucleotide-binding domain and leucine-rich repeat-containing receptor 3 (NLRP3), apoptosis-associated speck-like molecule containing a caspase recruitment domain (ASC), and caspase-1, but not on absent in melanoma 2 (AIM2), or gamma interferon-inducible protein 16 (IFI16). In contrast, HSV-1 activates non-canonical inflammasome signaling in proinflammatory macrophages that results in IL-1β, but not IL-18, release that is independent of NLRP3, ASC, and caspase-1. Ultraviolet irradiation of HSV-1 enhanced inflammasome activation, demonstrating that viral replication suppresses inflammasome activation. These results confirm that HSV-1 is capable of activating the inflammasome in human macrophages through an NLRP3 dependent process and that the virus has evolved an NLRP3 specific mechanism to inhibit inflammasome activation in macrophages.
Conflict of interest statement
The authors have declared that no competing interests exist.
Figures




References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous

