Distribution of functional opioid receptors in human dorsal root ganglion neurons
- PMID: 32102022
- PMCID: PMC7305987
- DOI: 10.1097/j.pain.0000000000001846
Distribution of functional opioid receptors in human dorsal root ganglion neurons
Abstract
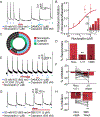
Preclinical evidence has highlighted the importance of the μ-opioid peptide (MOP) receptor on primary afferents for both the analgesic actions of MOP receptor agonists, as well as the development of tolerance, if not opioid-induced hyperalgesia. There is also growing interest in targeting other opioid peptide receptor subtypes (δ-opioid peptide [DOP], κ-opioid peptide [KOP], and nociceptin/orphanin-FQ opioid peptide [NOP]) on primary afferents, as alternatives to MOP receptors, which may not be associated with as many deleterious side effects. Nevertheless, results from several recent studies of human sensory neurons indicate that although there are many similarities between rodent and human sensory neurons, there may also be important differences. Thus, the purpose of this study was to assess the distribution of opioid receptor subtypes among human sensory neurons. A combination of pharmacology, patch-clamp electrophysiology, Ca imaging, and single-cell semiquantitative polymerase chain reaction was used. Our results suggest that functional MOP-like receptors are present in approximately 50% of human dorsal root ganglion neurons. δ-opioid peptide-like receptors were detected in a subpopulation largely overlapping that with MOP-like receptors. Furthermore, KOP-like and NOP-like receptors are detected in a large proportion (44% and 40%, respectively) of human dorsal root ganglion neurons with KOP receptors also overlapping with MOP receptors at a high rate (83%). Our data confirm that all 4 opioid receptor subtypes are present and functional in human sensory neurons, where the overlap of DOP, KOP, and NOP receptors with MOP receptors suggests that activation of these other opioid receptor subtypes may also have analgesic efficacy.
Figures






References
-
- Abdelhamid EE, Sultana M, Portoghese PS, Takemori AE. Selective blockage of delta opioid receptors prevents the development of morphine tolerance and dependence in mice. J Pharmacol Exp Ther 1991;258(1):299–303. - PubMed
-
- Anand P, Yiangou Y, Anand U, Mukerji G, Sinisi M, Fox M, McQuillan A, Quick T, Korchev YE, Hein P. Nociceptin/orphanin FQ receptor expression in clinical pain disorders and functional effects in cultured neurons. Pain 2016;157(9):1960–1969. - PubMed
-
- Bardoni R, Tawfik VL, Wang D, Francois A, Solorzano C, Shuster SA, Choudhury P, Betelli C, Cassidy C, Smith K, de Nooij JC, Mennicken F, O’Donnell D, Kieffer BL, Woodbury CJ, Basbaum AI, MacDermott AB, Scherrer G. Delta Opioid Receptors Presynaptically Regulate Cutaneous Mechanosensory Neuron Input to the Spinal Cord Dorsal Horn. Neuron 2014;81(6):1443. - PubMed
-
- Biggs JE, Yates JM, Loescher AR, Clayton NM, Boissonade FM, Robinson PP. Changes in vanilloid receptor 1 (TRPV1) expression following lingual nerve injury. Eur J Pain 2007;11(2):192–201. - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials

