Serum Levels and Removal by Haemodialysis and Haemodiafiltration of Tryptophan-Derived Uremic Toxins in ESKD Patients
- PMID: 32102247
- PMCID: PMC7073230
- DOI: 10.3390/ijms21041522
Serum Levels and Removal by Haemodialysis and Haemodiafiltration of Tryptophan-Derived Uremic Toxins in ESKD Patients
Abstract
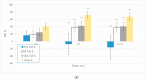
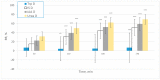
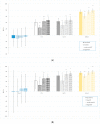
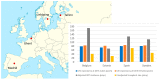
Tryptophan is an essential dietary amino acid that originates uremic toxins that contribute to end-stage kidney disease (ESKD) patient outcomes. We evaluated serum levels and removal during haemodialysis and haemodiafiltration of tryptophan and tryptophan-derived uremic toxins, indoxyl sulfate (IS) and indole acetic acid (IAA), in ESKD patients in different dialysis treatment settings. This prospective multicentre study in four European dialysis centres enrolled 78 patients with ESKD. Blood and spent dialysate samples obtained during dialysis were analysed with high-performance liquid chromatography to assess uremic solutes, their reduction ratio (RR) and total removed solute (TRS). Mean free serum tryptophan and IS concentrations increased, and concentration of IAA decreased over pre-dialysis levels (67%, 49%, -0.8%, respectively) during the first hour of dialysis. While mean serum total urea, IS and IAA concentrations decreased during dialysis (-72%, -39%, -43%, respectively), serum tryptophan levels increased, resulting in negative RR (-8%) towards the end of the dialysis session (p < 0.001), despite remarkable Trp losses in dialysate. RR and TRS values based on serum (total, free) and dialysate solute concentrations were lower for conventional low-flux dialysis (p < 0.001). High-efficiency haemodiafiltration resulted in 80% higher Trp losses than conventional low-flux dialysis, despite similar neutral Trp RR values. In conclusion, serum Trp concentrations and RR behave differently from uremic solutes IS, IAA and urea and Trp RR did not reflect dialysis Trp losses. Conventional low-flux dialysis may not adequately clear Trp-related uremic toxins while high efficiency haemodiafiltration increased Trp losses.
Keywords: chronic kidney disease; end-stage kidney disease; haemodiafiltration; haemodialysis; indole-3 acetic acid; indoxyl sulfate; tryptophan; tryptophan-derived uremic toxins; uremic toxins.
Conflict of interest statement
The authors declare no conflicts of interest. The funders had no role in the design of the study; in the collection, analyses, or interpretation of data; in the writing of the manuscript, or in the decision to publish the results.
Figures






Similar articles
-
Treatment with Paracetamol Can Interfere with the Intradialytic Optical Estimation in Spent Dialysate of Uric Acid but Not of Indoxyl Sulfate.Toxins (Basel). 2022 Sep 1;14(9):610. doi: 10.3390/toxins14090610. Toxins (Basel). 2022. PMID: 36136548 Free PMC article.
-
Protein-bound uraemic toxin removal in haemodialysis and post-dilution haemodiafiltration.Nephrol Dial Transplant. 2010 Jan;25(1):212-8. doi: 10.1093/ndt/gfp437. Epub 2009 Sep 15. Nephrol Dial Transplant. 2010. PMID: 19755476 Clinical Trial.
-
Indole 3-acetic acid, indoxyl sulfate and paracresyl-sulfate do not influence anemia parameters in hemodialysis patients.BMC Nephrol. 2017 Jul 26;18(1):251. doi: 10.1186/s12882-017-0668-5. BMC Nephrol. 2017. PMID: 28747155 Free PMC article.
-
Protein-bound uremic toxin lowering strategies in chronic kidney disease: a systematic review and meta-analysis.J Nephrol. 2021 Dec;34(6):1805-1817. doi: 10.1007/s40620-020-00955-2. Epub 2021 Jan 23. J Nephrol. 2021. PMID: 33484425
-
Hypoalbuminemia: a price worth paying for improved dialytic removal of middle-molecular-weight uremic toxins?Nephrol Dial Transplant. 2019 Jun 1;34(6):901-907. doi: 10.1093/ndt/gfy236. Nephrol Dial Transplant. 2019. PMID: 30102329 Review.
Cited by
-
The AKI-to-CKD Transition: The Role of Uremic Toxins.Int J Mol Sci. 2023 Nov 10;24(22):16152. doi: 10.3390/ijms242216152. Int J Mol Sci. 2023. PMID: 38003343 Free PMC article. Review.
-
Analysis of indoxyl sulfate in biological fluids with emphasis on sample preparation techniques: A comprehensive analytical review.Heliyon. 2024 Jul 23;10(15):e35032. doi: 10.1016/j.heliyon.2024.e35032. eCollection 2024 Aug 15. Heliyon. 2024. PMID: 39157307 Free PMC article. Review.
-
Treatment with Paracetamol Can Interfere with the Intradialytic Optical Estimation in Spent Dialysate of Uric Acid but Not of Indoxyl Sulfate.Toxins (Basel). 2022 Sep 1;14(9):610. doi: 10.3390/toxins14090610. Toxins (Basel). 2022. PMID: 36136548 Free PMC article.
-
Trained immunity: from kidney failure to organ transplantation.Nat Rev Nephrol. 2025 Apr;21(4):224-225. doi: 10.1038/s41581-024-00927-w. Nat Rev Nephrol. 2025. PMID: 39762368 No abstract available.
-
Indole-3-acetic acid correlates with monocyte-to-high-density lipoprotein (HDL) ratio (MHR) in chronic kidney disease patients.Int Urol Nephrol. 2022 Sep;54(9):2355-2364. doi: 10.1007/s11255-022-03137-0. Epub 2022 Feb 11. Int Urol Nephrol. 2022. PMID: 35147839
References
-
- Schmidt J.A., Rinaldi S., Scalbert A., Ferrari P., Achaintre D., Gunter M.J., Appleby P.N., Key T.J., Travis R.C. Plasma concentrations and intakes of amino acids in male meat-eaters, fish-eaters, vegetarians and vegans: A cross-sectional analysis in the epic-oxford cohort. Eur. J. Clin. Nutr. 2016;70:306–312. doi: 10.1038/ejcn.2015.144. - DOI - PMC - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical

