Comparison of the efficacy of MOE and PMO modifications of systemic antisense oligonucleotides in a severe SMA mouse model
- PMID: 32103257
- PMCID: PMC7102994
- DOI: 10.1093/nar/gkaa126
Comparison of the efficacy of MOE and PMO modifications of systemic antisense oligonucleotides in a severe SMA mouse model
Abstract
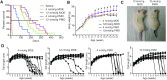
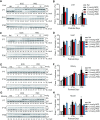
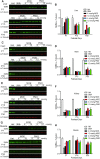
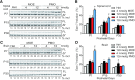
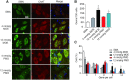
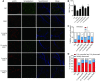
Spinal muscular atrophy (SMA) is a motor neuron disease. Nusinersen, a splice-switching antisense oligonucleotide (ASO), was the first approved drug to treat SMA. Based on prior preclinical studies, both 2'-O-methoxyethyl (MOE) with a phosphorothioate backbone and morpholino with a phosphorodiamidate backbone-with the same or extended target sequence as nusinersen-displayed efficient rescue of SMA mouse models. Here, we compared the therapeutic efficacy of these two modification chemistries in rescue of a severe mouse model using ASO10-29-a 2-nt longer version of nusinersen-via subcutaneous injection. Although both chemistries efficiently corrected SMN2 splicing in various tissues, restored motor function and improved the integrity of neuromuscular junctions, MOE-modified ASO10-29 (MOE10-29) was more efficacious than morpholino-modified ASO10-29 (PMO10-29) at the same molar dose, as seen by longer survival, greater body-weight gain and better preservation of motor neurons. Time-course analysis revealed that MOE10-29 had more persistent effects than PMO10-29. On the other hand, PMO10-29 appears to more readily cross an immature blood-brain barrier following systemic administration, showing more robust initial effects on SMN2 exon 7 inclusion, but less persistence in the central nervous system. We conclude that both modifications can be effective as splice-switching ASOs in the context of SMA and potentially other diseases, and discuss the advantages and disadvantages of each.
© The Author(s) 2020. Published by Oxford University Press on behalf of Nucleic Acids Research.
Figures


References
-
- De Clercq E., Eckstein E., Merigan T.C.. [Interferon induction increased through chemical modification of a synthetic polyribonucleotide]. Science (New York, N.Y.). 1969; 165:1137–1139. - PubMed
-
- Teplova M., Minasov G., Tereshko V., Inamati G.B., Cook P.D., Manoharan M., Egli M.. Crystal structure and improved antisense properties of 2′-O-(2-methoxyethyl)-RNA. Nat. Struct. Biol. 1999; 6:535–539. - PubMed
-
- Seth P.P., Siwkowski A., Allerson C.R., Vasquez G., Lee S., Prakash T.P., Kinberger G., Migawa M.T., Gaus H., Bhat B. et al.. Design, synthesis and evaluation of constrained methoxyethyl (cMOE) and constrained ethyl (cEt) nucleoside analogs. Nucleic Acids Symp. Ser. 2008; 2004:553–554. - PubMed
-
- Kumar R., Singh S.K., Koshkin A.A., Rajwanshi V.K., Meldgaard M., Wengel J.. The first analogues of LNA (locked nucleic acids): phosphorothioate-LNA and 2′-thio-LNA. Bioorg. Med. Chem. Lett. 1998; 8:2219–2222. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases
Research Materials

