Conformational Switching in Bcl-xL: Enabling Non-Canonic Inhibition of Apoptosis Involves Multiple Intermediates and Lipid Interactions
- PMID: 32111007
- PMCID: PMC7140517
- DOI: 10.3390/cells9030539
Conformational Switching in Bcl-xL: Enabling Non-Canonic Inhibition of Apoptosis Involves Multiple Intermediates and Lipid Interactions
Abstract
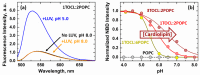
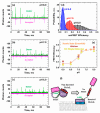
The inhibition of mitochondrial permeabilization by the anti-apoptotic protein Bcl-xL is crucial for cell survival and homeostasis. Its inhibitory role requires the partitioning of Bcl-xL to the mitochondrial outer membrane from an inactive state in the cytosol, leading to its extensive refolding. The molecular mechanisms behind these events and the resulting conformations in the bilayer are unclear, and different models have been proposed to explain them. In the most recently proposed non-canonical model, the active form of Bcl-xL employs its N-terminal BH4 helix to bind and block its pro-apoptotic target. Here, we used a combination of various spectroscopic techniques to study the release of the BH4 helix (α1) during the membrane insertion of Bcl-xL. This refolding was characterized by a gradual increase in helicity due to the lipid-dependent partitioning-coupled folding and formation of new helix αX (presumably in the originally disordered loop between helices α1 and α2). Notably, a comparison of various fluorescence and circular dichroism measurements suggested the presence of multiple Bcl-xL conformations in the bilayer. This conclusion was explicitly confirmed by single-molecule measurements of Fӧrster Resonance Energy Transfer from Alexa-Fluor-488-labeled Bcl-xL D189C to a mCherry fluorescent protein attached at the N-terminus. These measurements clearly indicated that the refolding of Bcl-xL in the bilayer is not a two-state transition and involves multiple membranous intermediates of variable compactness.
Keywords: BH4 domain; Bcl-2 proteins; Fluorescence Correlation Spectroscopy (FCS); Fluorescence Spectroscopy; Single Molecule FRET; apoptotic regulation; conformational switching; protein-membrane interactions.
Conflict of interest statement
The authors declare no conflict of interest.
Figures





References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials

