Structural analyses of the Group A flavin-dependent monooxygenase PieE reveal a sliding FAD cofactor conformation bridging OUT and IN conformations
- PMID: 32111738
- PMCID: PMC7136001
- DOI: 10.1074/jbc.RA119.011212
Structural analyses of the Group A flavin-dependent monooxygenase PieE reveal a sliding FAD cofactor conformation bridging OUT and IN conformations
Abstract
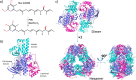
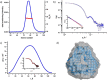
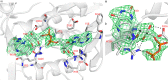
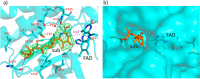
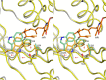
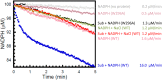
Group A flavin-dependent monooxygenases catalyze the cleavage of the oxygen-oxygen bond of dioxygen, followed by the incorporation of one oxygen atom into the substrate molecule with the aid of NADPH and FAD. These flavoenzymes play an important role in many biological processes, and their most distinct structural feature is the choreographed motions of flavin, which typically adopts two distinct conformations (OUT and IN) to fulfill its function. Notably, these enzymes seem to have evolved a delicate control system to avoid the futile cycle of NADPH oxidation and FAD reduction in the absence of substrate, but the molecular basis of this system remains elusive. Using protein crystallography, size-exclusion chromatography coupled to multi-angle light scattering (SEC-MALS), and small-angle X-ray scattering (SEC-SAXS) and activity assay, we report here a structural and biochemical characterization of PieE, a member of the Group A flavin-dependent monooxygenases involved in the biosynthesis of the antibiotic piericidin A1. This analysis revealed that PieE forms a unique hexamer. Moreover, we found, to the best of our knowledge for the first time, that in addition to the classical OUT and IN conformations, FAD possesses a "sliding" conformation that exists in between the OUT and IN conformations. This observation sheds light on the underlying mechanism of how the signal of substrate binding is transmitted to the FAD-binding site to efficiently initiate NADPH binding and FAD reduction. Our findings bridge a gap currently missing in the orchestrated order of chemical events catalyzed by this important class of enzymes.
Keywords: crystal structure; enzyme catalysis; enzyme mechanism; flavin adenine dinucleotide (FAD); flavin-dependent monooxygenase; hydroxylation; mobile flavin; piericidin; sliding conformation; small-angle X-ray scattering (SAXS).
© 2020 Manenda et al.
Conflict of interest statement
The authors declare that they have no conflicts of interest with the contents of this article
Figures


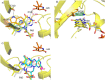

References
Publication types
MeSH terms
Substances
Associated data
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous

