BamA is required for autotransporter secretion
- PMID: 32114025
- PMCID: PMC7196024
- DOI: 10.1016/j.bbagen.2020.129581
BamA is required for autotransporter secretion
Abstract
Background: In Gram-negative bacteria, type Va and Vc autotransporters are proteins that contain both a secreted virulence factor (the "passenger" domain) and a β-barrel that aids its export. While it is known that the folding and insertion of the β-barrel domain utilize the β-barrel assembly machinery (BAM) complex, how the passenger domain is secreted and folded across the membrane remains to be determined. The hairpin model states that passenger domain secretion occurs independently through the fully-formed and membrane-inserted β-barrel domain via a hairpin folding intermediate. In contrast, the BamA-assisted model states that the passenger domain is secreted through a hybrid of BamA, the essential subunit of the BAM complex, and the β-barrel domain of the autotransporter.
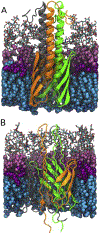
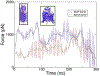
Methods: To ascertain the models' plausibility, we have used molecular dynamics to simulate passenger domain secretion for two autotransporters, EspP and YadA.
Results: We observed that each protein's β-barrel is unable to accommodate the secreting passenger domain in a hairpin configuration without major structural distortions. Additionally, the force required for secretion through EspP's β-barrel is more than that through the BamA β-barrel.
Conclusions: Secretion of autotransporters most likely occurs through an incompletely formed β-barrel domain of the autotransporter in conjunction with BamA.
General significance: Secretion of virulence factors is a process used by practically all pathogenic Gram-negative bacteria. Understanding this process is a necessary step towards limiting their infectious capacity.
Keywords: Autotransporters; Membrane proteins; Molecular dynamics; Secretion systems.
Copyright © 2020 Elsevier B.V. All rights reserved.
Figures
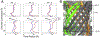


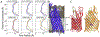

Similar articles
-
Job contenders: roles of the β-barrel assembly machinery and the translocation and assembly module in autotransporter secretion.Mol Microbiol. 2017 Nov;106(4):505-517. doi: 10.1111/mmi.13832. Epub 2017 Sep 26. Mol Microbiol. 2017. PMID: 28887826 Review.
-
Identification of a novel post-insertion step in the assembly of a bacterial outer membrane protein.Mol Microbiol. 2018 Oct;110(1):143-159. doi: 10.1111/mmi.14102. Epub 2018 Sep 28. Mol Microbiol. 2018. PMID: 30107065 Free PMC article.
-
Molecular basis for the folding of β-helical autotransporter passenger domains.Nat Commun. 2018 Apr 11;9(1):1395. doi: 10.1038/s41467-018-03593-2. Nat Commun. 2018. PMID: 29643377 Free PMC article.
-
BamA forms a translocation channel for polypeptide export across the bacterial outer membrane.Mol Cell. 2021 May 6;81(9):2000-2012.e3. doi: 10.1016/j.molcel.2021.02.023. Epub 2021 Mar 10. Mol Cell. 2021. PMID: 33705710 Free PMC article.
-
Looks can be deceiving: recent insights into the mechanism of protein secretion by the autotransporter pathway.Mol Microbiol. 2015 Jul;97(2):205-15. doi: 10.1111/mmi.13031. Epub 2015 May 15. Mol Microbiol. 2015. PMID: 25881492 Free PMC article. Review.
Cited by
-
ATP-independent assembly machinery of bacterial outer membranes: BAM complex structure and function set the stage for next-generation therapeutics.Protein Sci. 2024 Feb;33(2):e4896. doi: 10.1002/pro.4896. Protein Sci. 2024. PMID: 38284489 Free PMC article. Review.
-
TamAB is regulated by PhoPQ and functions in outer membrane homeostasis during Salmonella pathogenesis.J Bacteriol. 2023 Oct 26;205(10):e0018323. doi: 10.1128/jb.00183-23. Epub 2023 Sep 20. J Bacteriol. 2023. PMID: 37728604 Free PMC article.
-
Folding Control in the Path of Type 5 Secretion.Toxins (Basel). 2021 May 11;13(5):341. doi: 10.3390/toxins13050341. Toxins (Basel). 2021. PMID: 34064645 Free PMC article. Review.
-
Uncovering the folding mechanism of pertactin: A comparative study of isolated and vectorial folding.Biophys J. 2023 Jul 25;122(14):2988-2995. doi: 10.1016/j.bpj.2023.03.021. Epub 2023 Mar 22. Biophys J. 2023. PMID: 36960532 Free PMC article.
-
Examining Protein Translocation by β-Barrel Membrane Proteins Using Reconstituted Proteoliposomes.Methods Mol Biol. 2024;2778:83-99. doi: 10.1007/978-1-0716-3734-0_6. Methods Mol Biol. 2024. PMID: 38478273
References
-
- van den Berg B, Crystal structure of a full-length autotransporter, J. Mol. Biol 396 (2010) 627–633. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous

