Indirect Impact of PD-1/PD-L1 Blockade on a Murine Model of NK Cell Exhaustion
- PMID: 32117218
- PMCID: PMC7026672
- DOI: 10.3389/fimmu.2020.00007
Indirect Impact of PD-1/PD-L1 Blockade on a Murine Model of NK Cell Exhaustion
Abstract
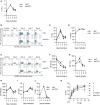
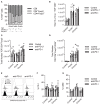
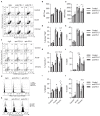
The induction of exhaustion on effector immune cells is an important limiting factor for cancer immunotherapy efficacy as these cells undergo a hierarchical loss of proliferation and cytolytic activity due to chronic stimulation. Targeting PD-1 has shown unprecedented clinical benefits for many cancers, which have been attributed to the prevention of immune suppression and exhaustion with enhanced anti-tumor responses. In this study, we sought to evaluate the role of the PD-1/PD-L1 pathway in murine natural killer (NK) cell activation, function, and exhaustion. In an in vivo IL-2-dependent exhaustion mouse model, neutralization of the PD-1/PD-L1 pathway improved NK cell activation after chronic stimulation when compared to control-treated mice. These cells displayed higher proliferative capabilities and enhanced granzyme B production. However, the blockade of these molecules during long-term in vitro IL-2 stimulation did not alter the progression of NK cell exhaustion (NCE), suggesting an indirect involvement of PD-1/PD-L1 on NCE. Given the expansion of CD8 T cells and regulatory T cells (Tregs) observed upon acute and chronic stimulation with IL-2, either of these two populations could influence NK cell homeostasis after PD-L1/PD-1 therapy. Importantly, CD8 T cell activation and functional phenotype were indeed enhanced by PD-1/PD-L1 therapy, particularly with anti-PD-1 treatment that resulted in the highest upregulation of CD25 during chronic stimulation and granted an advantage for IL-2 over NK cells. These results indicate a competition for resources between NK and CD8 T cells that arguably delays the onset of NCE rather than improving its activation during chronic stimulation. Supporting this notion, the depletion of CD8 T cells reversed the benefits of PD-1 therapy on chronically stimulated NK cells. These data suggest a bystander effect of anti-PD1 on NK cells, resulting from the global competition that exists between NK and CD8 T cells for IL-2 as a key regulator of these cells' activation. Thus, achieving an equilibrium between these immune cells might be important to accomplish long-term efficacy during anti-PD-1/IL-2 therapy.
Keywords: CD8; NK; PD-1/PD-L1 pathway; chronic stimulation; exhaustion.
Copyright © 2020 Alvarez, Simonetta, Baker, Morrison, Wenokur, Pierini, Berraondo and Negrin.
Figures




Similar articles
-
PD-L1 blockade synergizes with IL-2 therapy in reinvigorating exhausted T cells.J Clin Invest. 2013 Jun;123(6):2604-15. doi: 10.1172/JCI67008. Epub 2013 May 15. J Clin Invest. 2013. PMID: 23676462 Free PMC article.
-
Sensitizing tumors to anti-PD-1 therapy by promoting NK and CD8+ T cells via pharmacological activation of FOXO3.J Immunother Cancer. 2021 Dec;9(12):e002772. doi: 10.1136/jitc-2021-002772. J Immunother Cancer. 2021. PMID: 34887262 Free PMC article.
-
PD-L1 targeting high-affinity NK (t-haNK) cells induce direct antitumor effects and target suppressive MDSC populations.J Immunother Cancer. 2020 May;8(1):e000450. doi: 10.1136/jitc-2019-000450. J Immunother Cancer. 2020. PMID: 32439799 Free PMC article.
-
What Do We Have to Know about PD-L1 Expression in Prostate Cancer? A Systematic Literature Review. Part 3: PD-L1, Intracellular Signaling Pathways and Tumor Microenvironment.Int J Mol Sci. 2021 Nov 15;22(22):12330. doi: 10.3390/ijms222212330. Int J Mol Sci. 2021. PMID: 34830209 Free PMC article.
-
Immunotherapy for head and neck cancers: an update and future perspectives.Immunotherapy. 2019 May;11(7):561-564. doi: 10.2217/imt-2019-0022. Immunotherapy. 2019. PMID: 30943860 Review. No abstract available.
Cited by
-
Targeting Triple NK Cell Suppression Mechanisms: A Comprehensive Review of Biomarkers in Pancreatic Cancer Therapy.Int J Mol Sci. 2025 Jan 9;26(2):515. doi: 10.3390/ijms26020515. Int J Mol Sci. 2025. PMID: 39859231 Free PMC article. Review.
-
Exploration of a Robust and Prognostic Immune Related Gene Signature for Cervical Squamous Cell Carcinoma.Front Mol Biosci. 2021 Mar 3;8:625470. doi: 10.3389/fmolb.2021.625470. eCollection 2021. Front Mol Biosci. 2021. PMID: 33748188 Free PMC article.
-
The cancer-natural killer cell immunity cycle.Nat Rev Cancer. 2020 Aug;20(8):437-454. doi: 10.1038/s41568-020-0272-z. Epub 2020 Jun 24. Nat Rev Cancer. 2020. PMID: 32581320 Review.
-
Cystatin A promotes the antitumor activity of T helper type 1 cells and dendritic cells in murine models of pancreatic cancer.Mol Oncol. 2025 May;19(5):1452-1470. doi: 10.1002/1878-0261.13796. Epub 2025 Jan 10. Mol Oncol. 2025. PMID: 39792573 Free PMC article.
-
What Happens to the Immune Microenvironment After PD-1 Inhibitor Therapy?Front Immunol. 2021 Dec 23;12:773168. doi: 10.3389/fimmu.2021.773168. eCollection 2021. Front Immunol. 2021. PMID: 35003090 Free PMC article. Review.
References
-
- Melero I, Rouzaut A, Motz GT, Coukos G. T-cell and NK-cell infiltration into solid tumors: a key limiting factor for efficacious cancer immunotherapy. Cancer Discov. (2014) 4:522–6. 10.1158/2159-8290.CD-13-0985 - DOI - PMC - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials

